Introduction
Chronic venous leg ulcers (VLUs) are one of the most common forms of chronic lower limb wounds, accounting for approximately 60-80% of all leg ulcers and affecting 1-3% of the adult population in developed nations.1 The prevalence increases to nearly 5% among individuals aged 65 and older, with further increases projected as populations age.1,2 In the UK, the economic burden of chronic leg ulcer care is considerable, estimated at £1.9 billion annually,3 with chronic venous ulcer treatment costs estimated to be around £941 million.4 The high costs are associated with frequent clinic visits, extended dressing care, compression therapy, and related hospitalizations.2,3 VLUs profoundly impair patients' mobility, quality of life, and productivity due to discomfort, odor, social isolation, and the risk of repeated recurrences.5,6 With ageing populations, this cost is likely to rise further.
Along with systemic factors, reduced mobility, inadequate ankle function, and poor adherence to compression therapy are the primary factors contributing to delayed healing. Even with good debridement and compression therapy, almost 20-30% of chronic venous ulcers do not heal, further adding to the socio-economic burden.4,7 Characteristics specific to the wound, such as its size (over 8.25 cm2), depth, and location, have also proven reliable indicators of prognosis.1 Wounds that are larger and deeper (>2 cm deep) exhibit a greater inflammatory burden and disrupted skin repair.1,7 Similarly, a larger calf circumference and a long ulcer duration make healing more challenging.8
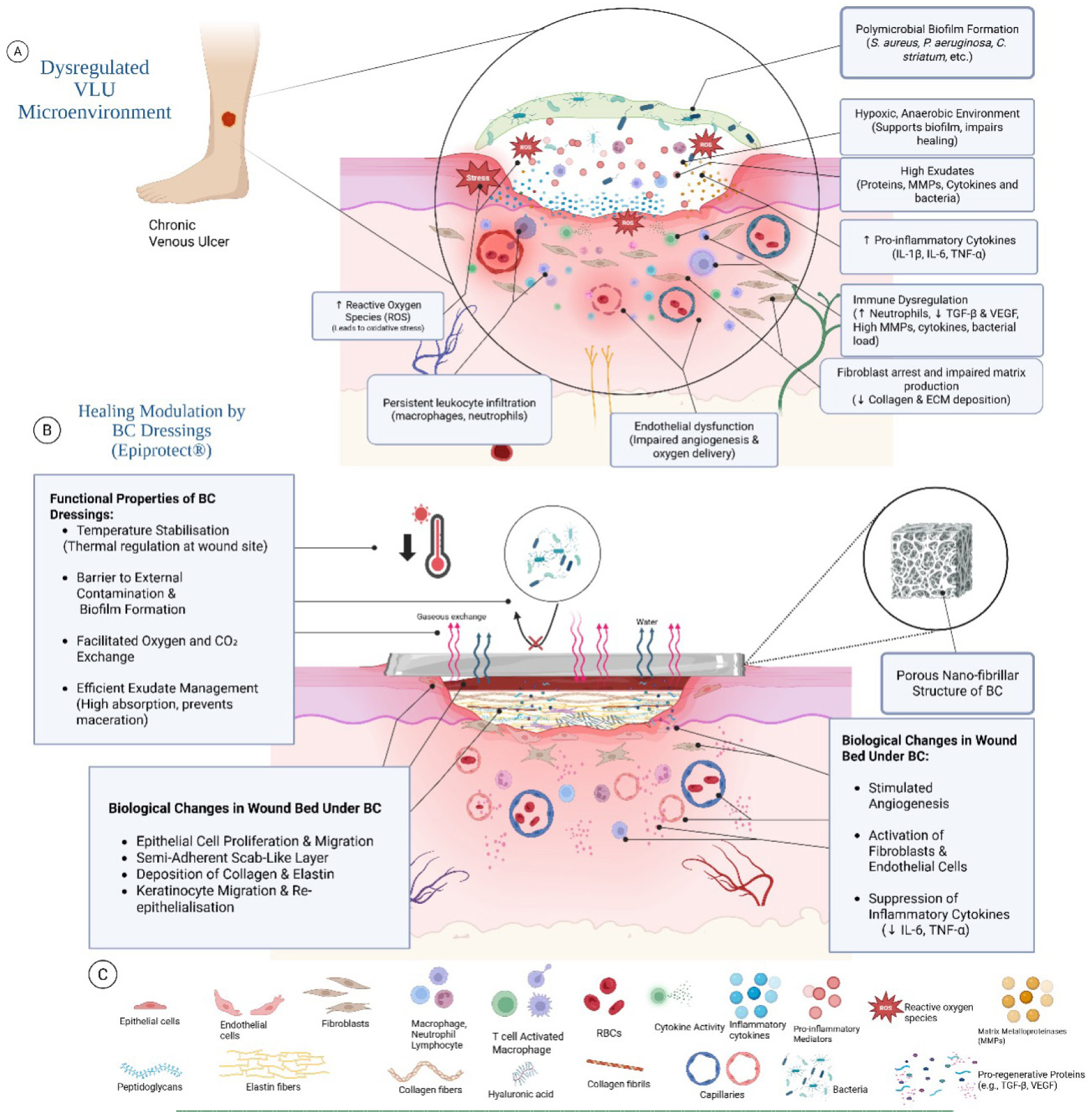
Biosynthetic cellulose (BC) is a natural dressing that meets the requirements of a modern active wound dressing, which is biocompatible, non-toxic, and easy to use and store.9 It has a three-dimensional network of 3D nanofilaments that can potentially prevent biofilm formation and external microbial infections. The porous three-dimensional structure of BC enables gas and liquid exchange, promoting rapid wound healing. Due to its chemical nature, it also exhibits strong water absorption and retention, which supports the absorption of exudates.10–12 BC dressings have been proposed to support wound healing by maintaining a moist and thermally stable wound microenvironment, driven by their high water-binding capacity and nanofibrillar hydrogel structure, which may reduce evaporative heat loss and support cellular migration and enzymatic activity at the wound surface.13 Once the healing is complete, the dressing falls off like a scab, leaving no remnants behind (Figure 1).14
FIGURE 1 Illustration of the chronic wound microenvironment in venous leg ulcers (VLUS) and the proposed mechanisms by which biosynthetic cellulose (BC) dressings, such as Epiprotect, promote healing. The top panel highlights key features of VLU pathophysiology, including biofilm formation, endothelial dysfunction, and inflammatory dysregulation. The lower panel illustrates how BC supports wound healing by modulating inflammation, managing exudate, promoting tissue regeneration, and stabilising the microenvironment. Temperature stabilisation involves BC dressings maintaining a moist, thermally stable wound environment, potentially reducing heat loss and supporting cellular functions activity; Molecular and cellular components are indicated in the legend below.13,15-20 Illustration created in https://biorender.com
Epiprotect Ulcer is a new alternative BC dressing sheet manufactured using Eiratex, a proprietary exopolysaccharide developed by S2Medical AB (Sweden). The biomaterial is identical to a plant-derived cellulose (β-1-4-glucose), devoid of lignin and hemicellulose. Exopolysaccharide is produced and secreted by aerobic gram-negative bacteria Komagataeibacter xylinus (formerly known as Gluconabacter xylinus). BC forms a hydrated and entangled 3D network of well-defined nanocellulose fibrils.21-23
Epiprotect shows promise across various patient groups, including adults with burns, children, and the elderly with chronic wounds. In a Swedish trial, Epiprotect and porcine xenografts for adult partial-thickness burns showed similar healing, pain, infection rates, costs, and scarring outcomes. However, patients rated their scars worse at one year with porcine dressings, despite clinicians not noticing a difference.24 The benefits were more notable among children. Qureshi et al25 found Epiprotect reduced pain, anxiety, and hypertrophic scars compared to gauze, with similar healing times. A UK study using Epiprotect in children under 5 years old confirmed that it was safe and well-tolerated, and that it prevented painful dressing changes. Most healed quickly without needing skin grafts.26 Similar results were seen in a pilot study by Delli Santi and Borgognone.27
In a case series of elderly patients with non-healing ulcers, Epiprotect Ulcer aided in closing chronic wounds with fewer than two dressings per patient over 6 weeks.28 Research by Sivlér et al shows that Epiprotect effectively heals ulcers with minimal dressing changes.28
This service evaluation study aimed to assess Epiprotect's clinical effectiveness in managing chronic VLUs in the UK, focusing on wound healing, patient outcomes, and dressing performance, given the lack of robust clinical studies in this area.
Methodology
This was a prospective, single-arm, self-controlled service evaluation of the use of Epiprotect in the treatment of chronic VLUs at the Pioneer Wound Healing and Lymphoedema Centers (WHLC). Patients from two centers of this clinic (Crawley and Eastbourne) were enrolled in this study. The study took place at these centers between February 2025 and August 2025. Each participant served as their own control, receiving standard care (according to Pioneer WHLC clinic and National Institute for Health and Care Excellence (NICE) guidelines)29,30, followed by crossover to Epiprotect treatment. The local clinic and UCL (DSIS) approved this project. Research procedures were conducted in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Declaration of Helsinki of 1975, as amended in 2013. Since this study was a service evaluation, it was exempted from UCL or NIHR Research Ethics Committee approval and registration.
Participants
A total of 20 adult participants with a confirmed diagnosis of chronic venous ulcers (CVUs) (based on a detailed history, primary wound assessment, NHS referrals, and vascular assessment) were enrolled in this study. Informed consent was obtained before enrolling the patients, in accordance with CGP guidelines.31 The study clinician and research nurses at Pioneer WHLC assessed eligibility. Inclusion and exclusion criteria for this service evaluation were as follows:
Inclusion criteria
-
Age ≥18 years
-
VLU duration >6 weeks
-
Wound area <200 cm2
-
No clinical signs of infection
-
Adherence to comorbidity treatment.
Exclusion criteria
-
Known allergies to trial dressings or compression therapy
-
Antibiotic use within 7 days before enrolment
-
Prolonged use of corticosteroids or immunosuppressants
-
Comorbidities affecting wound healing (e.g., autoimmune disease, cellulitis)
-
Participation in another wound-healing study within 30 days
-
History of non-adherence to treatment.
Study design and protocol
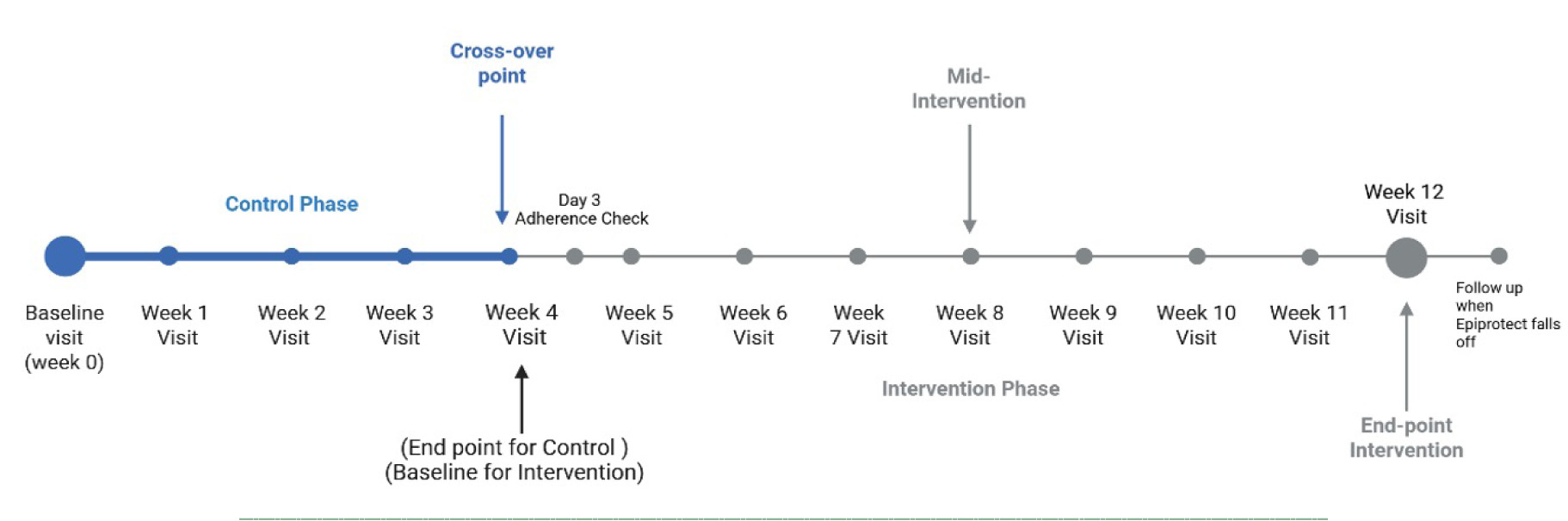
The study lasted 12 weeks. At week 0, baseline measurements were taken after cleaning and, if needed, debridement. During the control phase (weeks 0-4), patients were visited at least three times. All leg ulcers received compression.
At week 4, the wound was cleaned, and debridement was performed if necessary (standard of care); then Epiprotect was applied. Patients visited the clinic on day 3 post-application to check dressing adherence. They then returned weekly for 8 weeks for measurements. After 12 weeks, if Epiprotect remained adherent, they were asked to follow up once it detached naturally.
On each visit, the dressings were changed to assess exudate staining, pain scores were recorded, and photographs of the wound after cleaning were taken (Figure 2).
FIGURE 2 Timeline and phases of this self-controlled study. Created in https://biorender.com
Materials used
During the control phase, dressing materials included moist gauze, super-absorbent (Zetuvit, Hartmann, Germany) dressing, and two-layer compression (Actico 2-layer, L&R, Germany). During the intervention, the dressing was Epiprotect, gauze, super-absorbent dressing (used for the 1st week or replaced with new Epiprotect), and two-layer compression.
Data collection tools
Patient data and weekly wound images were stored in the Pioneer WHLC Digital Database (using JellyPx PIMS). Wound measurements were taken using Dermicus (Sweden)32,33, a mobile imaging application, and confirmed manually using ImageJ (National Institutes of Health (NIH), US).34 Calibration measurements were taken using in-photo rulers by two blinded assessors. Any discrepancies exceeding 10% were resolved through consensus. The final data were then compiled in Microsoft Excel (Microsoft Corp., US) for additional analysis.
All data were anonymized using study IDs and securely stored in password-protected research databases—only authorized personnel accessed sensitive data in compliance with NHS governance protocols.31
Outcomes: definition and measurement
The following outcomes were measured.
Primary outcomes
Percentage of wound area reduction (PWAR): is defined as the relative percentage change from the original wound size.35
PWAR=(Area Baseline-Area Observed)/(Area Baseline) X 100
The area baseline for control is week 0 area, and the area baseline for intervention is week 4 area. (area baseline = baseline area for phase; area observed = Area of week n)
Healing thresholds (≥50%, ≥75%, ≥90%, and 100%) and the time to achieve them were also recorded.36
Secondary outcomes
• Pain: subjective pain rating reported by patient on a 0-10 scale. Recorded weekly visual analogue scale score (0 = no pain, 10 = worst pain).37 Pain scores were measured at enrollment, the end of the control phase, and weekly after applying Epiprotect.37
-
Total number of Epiprotect sheets used: total BC sheets used during intervention and sheets per patient.
-
Adherence rate: observed percentage of BC sheet adhered to the wound bed.38,39 Recorded on day 3 post-intervention and weekly thereafter. Reapplications were also noted.
-
Exudate score: grade weekly dressing stain using a non-validated visual scale. Two independent scorers graded exudate level weekly on a 0-3 scale, resolving discrepancies by consensus (Table 1).
-
Patient demographics: age, sex, height, weight, body mass index (BMI), smoking status (cigarettes/day), weekly alcohol consumption (units/week), co-morbidities, and medication history recorded on the day of enrolment in the study.
-
Ulcer characteristics: duration of ulcer (>6 months, <6months), location of ulcer, and ulcer classification (CEAP classification).40
TABLE 1 A 4-point clinician-reported Exudate Score was developed for this study
| Score | Grading | Inference |
|---|---|---|
| Score 0 | None | Dressing dry, no visible moisture. |
| Score 1 | Scant | Light exudate, less than 25% of dressing stained. |
| Score 2 | Moderate | Moist dressing, 25-75% of dressing stained. |
| Score 3 | Heavy | Dressing saturated or leaking, more than 75% stained or with strike-through. |
Adapted from elements of the PUSH Tool and Bates-Jensen Wound Assessment Tool (BWAT), but designed to assess dressing stain patterns during weekly dressing changes.
Descriptive statistics were calculated, and normality was tested with the Shapiro-Wilk test. Data were shown as mean ± standard deviation (SD) or median (interquartile range (IQR)). Significance was p<0.05, confidence interval (CI) >95%. Analyses were conducted using SPSS v29 (IBM Corp., US) and Python (Python Software Foundation, US) packages.
PWAR was selected for its widespread use in venous ulcer research as a relative healing metric but was interpreted alongside absolute wound area reduction and healing thresholds to mitigate the known volatility of small, heterogeneous wounds or those exhibiting early deterioration. Primary analyses were pre-specified as comparisons of PWAR and absolute wound area reduction across defined study phases (control, intervention, follow-up). Week-to-week comparisons, correlation analyses, and secondary outcomes (pain, exudate, adherence) were considered exploratory and hypothesis-generating. No formal correction for multiple testing was applied due to the small sample size and service-evaluation design, where such adjustment would substantially increase the risk of type II error.
Analysis: population and missing data handling
A case-available sensitivity analysis was conducted using the per-protocol subset, which included participants up to their last valid observation before deviation or dropout. In the “censor-at-deviation” approach, only data before the deviation were included, excluding subsequent measurements. This ensured each participant contributed valid control and early-intervention data despite incomplete follow-up. Analyses were conducted using this approach, including all participants who contributed valid data up to their last available observation. No imputation of missing data was performed; analyses were conducted using available observations at each time point. Correlation analyses were restricted to complete cases. Given the small sample size and non-normal distribution of several outcomes, non-parametric bootstrapping was used to support inferential analyses. All primary and secondary statistical tests were bootstrapped using 1000 resamples with replacement to estimate confidence intervals and assess the stability of effect estimates. Bootstrapping was applied as a robustness check and did not replace the primary analytical framework. Dropout information and reasons for withdrawal are detailed in Supplementary Table 1.
Results
Overview of data
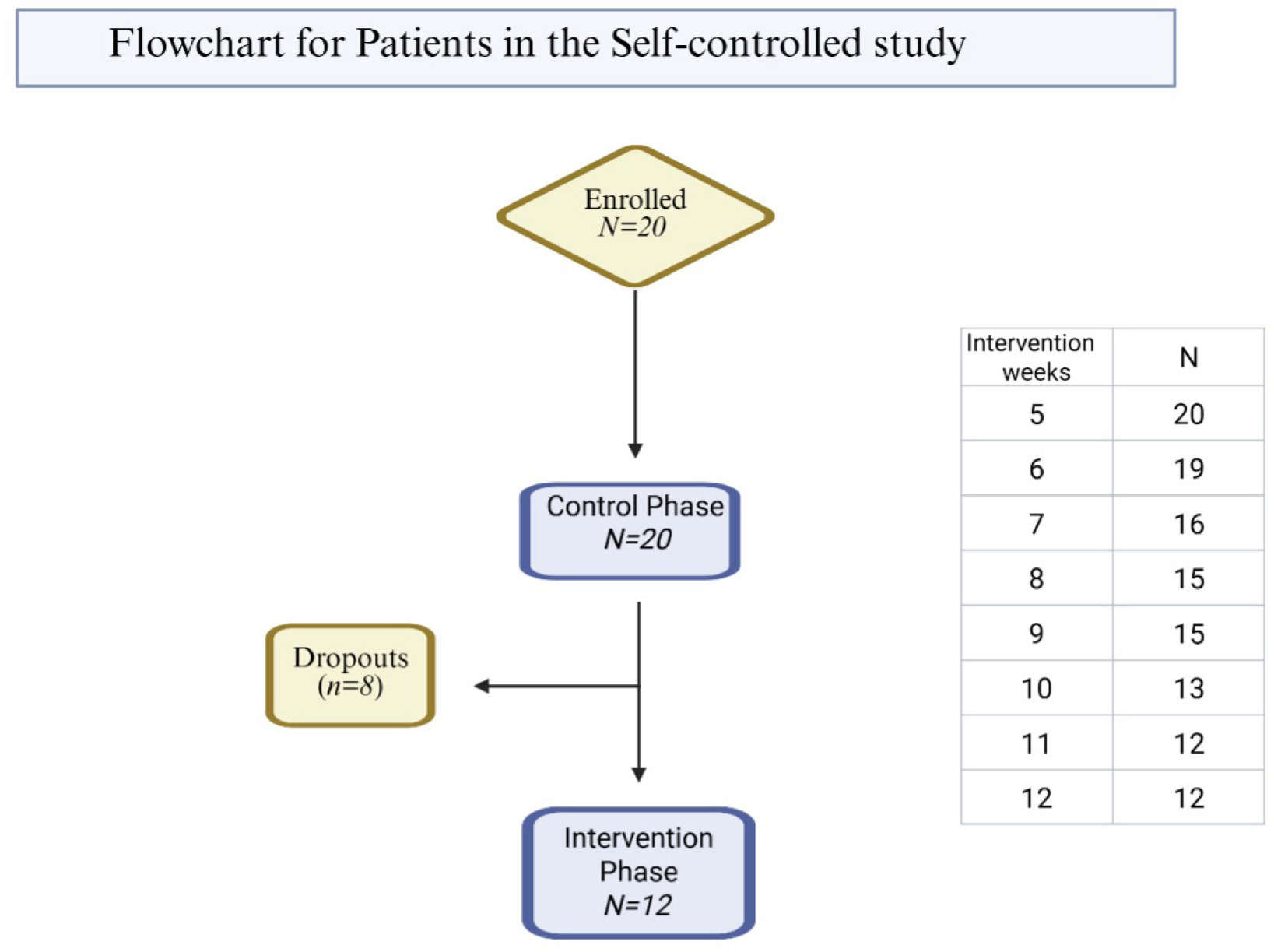
The control phase included 20 participants, of whom 12 completed the study by its conclusion. The dataset included 12 participants with complete 12-week data and 8 participants with partial data up to their last credible point, resulting in an analysis cohort of 20 participants contributing data up to their last valid observation. All primary and secondary statistical tests included non-parametric tests (Wilcoxon signed rank and Friedman) and a parametric test (paired t). Correlation analyses used only complete cases to avoid bias introduced by imputation. To account for the small sample size and incomplete follow-up, primary and secondary analyses were supported by non-parametric bootstrapping, which confirmed the stability and direction of observed effects. The results remained consistent in terms of direction, supporting the robustness of the primary findings (Figure 3 and Supplementary Table 1).
FIGURE 3 Flowchart of patient inclusion and progression through study phases.
Participant characteristics
The mean age of the participants was 68.6 years (range, 22-93), with an equal gender distribution (10 males and 10 females). The mean BMI was 34.3±10.7 kg/m2. Most of the ulcers were CEAP Grade C6 active (n=18), with a minority classified as C6 recurrent (n=2). Common comorbidities included hypertension (n=9) and type 2 diabetes mellitus (n=5). The participants' ethnicity was predominantly Caucasian (n=18) and South Asian (n=2). Detailed demographic data are presented in Table 2.
TABLE 2 Demographic data of the participants
| Characteristics | Value |
|---|---|
| Number of patients | N=20 |
| Age (years) | Mean: 68.6 (Range: 22-93) |
| Gender | 10 Male, 10 Female |
| Height (m) | Mean: 1.73 ± 0.11 |
| Weight (kg) | Mean: 104.2 ± 38.3 |
| BMI (kg/m2) | Mean: 34.3 ± 10.7 |
| Smoking status | The majority never smoked; n=3 ex-smokers |
| Alcohol use | Light use in a few (2-5 units/week) |
| Ulcer duration | Range: Feb 2021 - Dec 2024 |
| Ulcer duration, n (%) | >6 months: n=13 (65%) ≤6 months: n=6 (30%), Unknown: n=1 (5%) |
| CEAP grade | N=18, C6 (active), N=2, C6 (recurrent) |
| Ulcer size (baseline) | 7.3±10.24 cm2 |
| Wound location | R anterior gaiter (n=5) L anterior gaiter (n=3) L medial gaiter (n=3) Lateral gaiter (n=4) R posterior gaiter (n=1) L posterior gaiter (n=1) L lower circumferential (n=1) L medial malleolus (n=2) |
| Co-morbidities | Hypertension (n=9) Type 2 Diabetes (n=5) Osteoarthritis (n=2) Heart disease (n=2) Anaemia (n=2) CKD (n=2) (multimorbidity present) |
| Medications | Frequent use of antihypertensives, statins, diuretics, and antidiabetics |
| Nutritional/bone health support | Agents such as alendronic acid are noted in elderly female patients |
Primary outcome
Percentage wound area reduction (PWAR)
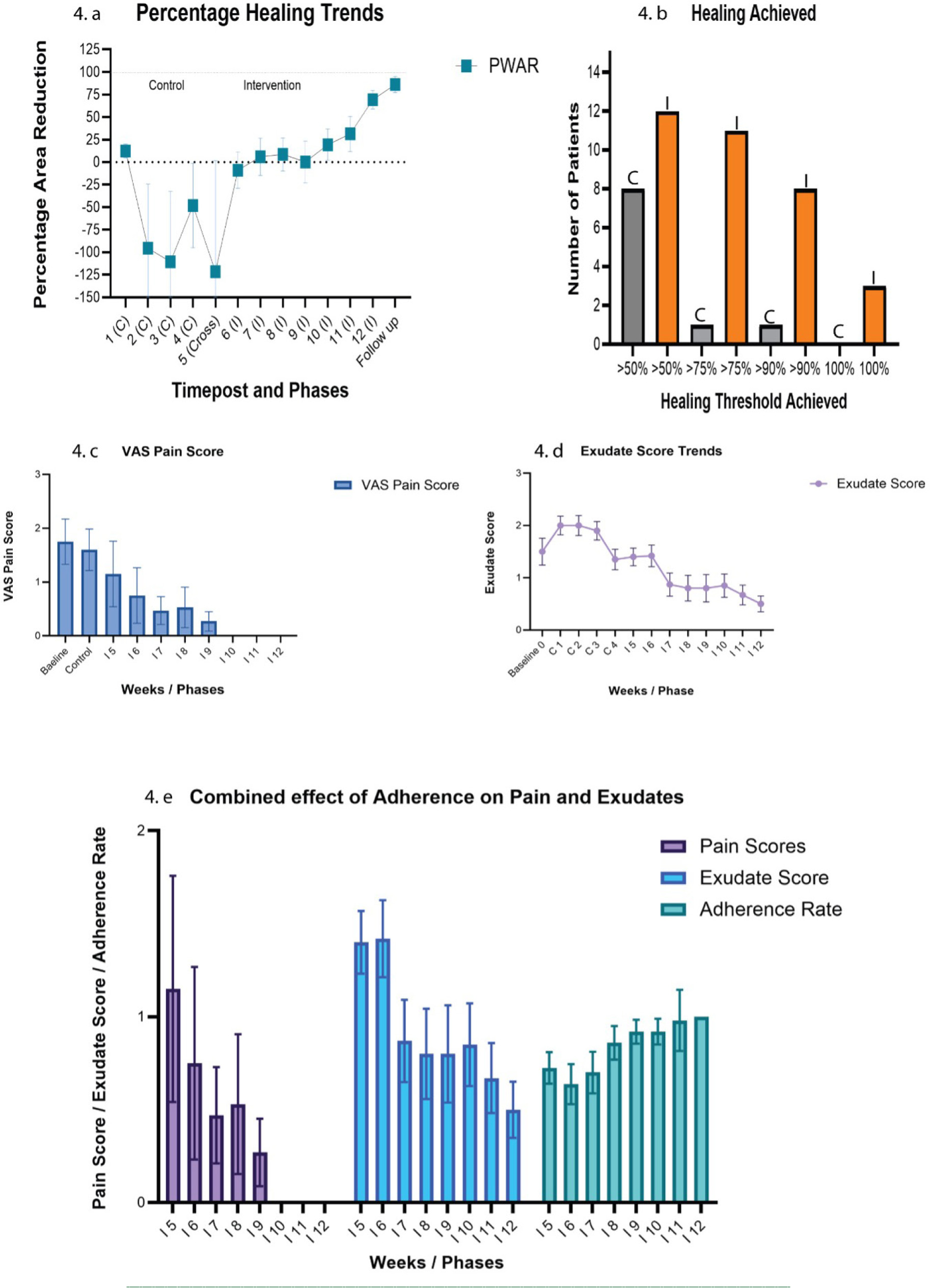
PWAR values varied during control (weeks 1–4), rising from 12.45±34.18% at week 1 to −48.10±209.49% at week 4. After starting Epiprotect, values improved from −121.47±549.99% at week 5 to 86.11±31.11% at follow-up. There was a significant increase in PWAR from week 4 (control) to week 12 (intervention) (p=0.046/0.015; r=0.70), with a large effect size. The increase in PWAR persisted at follow-up (p=0.017/0.002; r=0.88), indicating sustained improvement. A strong time-dependent association (p<0.001) was observed, with progressive healing during the Epiprotect phase. Absolute area reduction dropped from 7.36±10.22 cm2 at baseline to 0.50±1.12 cm2 at follow-up (week 12 (p=0.004) and follow-up (p=0.003), with large effect sizes (r=0.58–0.63)) (See Figure 4 and Supplementary Tables 2 and 3).
FIGURE 4 Healing, pain, exudate, and adherence outcomes during Epiprotect intervention. a) PWAR increased steadily after Epiprotect introduction, with significant improvements by week 12 (p=0.046, r=0.70) and follow-up (p=0.002, r=0.88). b) Kaplan– Meier analysis showed faster progression to ≥75 % and ≥90 % healing (median 7 and 12 weeks, respectively), with three complete closures under Epiprotect. c) VAS pain scores declined significantly (χ2 = 38.15, p<0.001), showing large–very large effects from week 6 onward. d) Exudate scores fell markedly (χ2 = 69.85, p<0.001), indicating improved wound environment. e) Adherence improved over time (χ2 = 20.45, p=0.006) and correlated positively with PWAR (r = 0.62) and inversely with pain and exudate (r ≈ –0.6). Collectively, these results demonstrate enhanced healing dynamics and patient comfort with Epiprotect.
Healing threshold
At ≥50%, 8% of controls and 40% of Epiprotect wounds healed, with mean healing times of 3.20 and 6.11 weeks. For ≥75%, in 5% of controls and 55% of Epiprotect wounds this was achieved, with mean times of 3.9 and 10 weeks, respectively. At a rate of greater than 90%, 40% of Epiprotect and 5% of controls reached this level, with a mean time of 10.84 weeks. Complete healing occurred in 15% of Epiprotect wounds, but none of the controls (Figure 4 and Supplementary Table 4).
Pain scores (VAS)
Mean pain scores decreased from 1.75±1.89 at baseline and 1.60±1.73 during control to 1.15±2.72 by week 5, 0.53±1.46 by week 8, and near zero by week 12. There was no difference between baseline and control (p=0.53; r=0.14) but a significant reduction from control to week 6 (p=0.027; r=0.50), continuing through weeks 7 (p=0.04; r=0.50), 9 (p=0.02; r=0.60), and 10–12 (p=0.02–0.006; r=0.60–0.80). A significant overall reduction (p<0.001) was observed (Figure 4 and Supplementary Tables 2 and 5).
Exudate control
Exudate levels decreased from 1.50±1.15 at baseline to 0.50±0.52 at week 12. Early comparisons (baseline–week 4) were non-significant (p=0.74), but reductions became significant: week 5–week 1 (p=0.007; r=0.60), week 6–week 2 (p=0.008; r=0.61), and week 7–week 3 (p=0.001; r=0.81). The late intervention phase showed significant differences relative to the control (week 4) at weeks 11 and 12 (p=0.053–0.030; r=0.56–0.63). A significant overall reduction (p<0.001) was observed (Figure 4 and Supplementary Tables 2 and 5).
Adherence and dressing use
Adherence rates were high throughout the intervention and improved significantly over time (p=0.006). Mean adherence increased from 78±36.9% on day 3 to 86.7±35.2% on week 8 and 91±10.8% on week 12 (Figure 4 and Supplementary Table 5).
The average number of sheets used during the intervention was low, at 2.20 ± 1.25 dressings per participant (range, 1–6) (Figure 4 and Supplementary Tables 2 and 5).
Correlation analyses
Strong correlations were found between adherence to the BC sheet and healing metrics. Early day 3 adherence correlates with smaller final wound sizes (r=−0.63, p<0.001) and greater 12-week area reduction (r=0.62, p<0.001). It also shows an inverse relationship with exudate (r=−0.46, p<0.001). Intervention adherence is negatively correlated with 12-week wound size (r=−0.30, p<0.001) and exudate (r=−0.69, p<0.001), and weakly positively correlated with PWAR (r=0.24, p<0.001) (Supplementary Table 4).
Visual analysis of wound progression
Serial images demonstrated progressive epithelialization, contraction, reduction in slough and exudate, and improved peri-ulcer skin after Epiprotect. Most ulcers exhibited a clear transition from an inflammatory to a reparative appearance by mid-intervention, with stable epithelial coverage at follow-up (Figures 5 and 6).
FIGURE 5 Representative wound healing trajectories during the control and Epiprotect intervention phase. Images were acquired under consistent lighting and distance using Dermicus® digital imaging. Sequential clinical photographs from three patients illustrate progressive wound contraction and epithelialisation across study phases (Baseline → Control → Mid-intervention → Intervention End → Follow-up). All wounds were chronic venous leg ulcers located on the gaiter region. Each row represents an individual case: Top row (Patient 1001, M, 72 y): PWAR improved from 40.47 % (control) to 98 % at follow-up, with near-complete closure by Week 12; Middle row (Patient 1003, M, 63 y): PWAR increased from 43.13 % to 96.24 %, showing rapid granulation and epithelial edge migration after Epiprotect application; Bottom row (Patient 1013, M, 87 y): PWAR rose from 49.10 % to 96.37 %, demonstrating sustained reduction in wound area and inflammation.
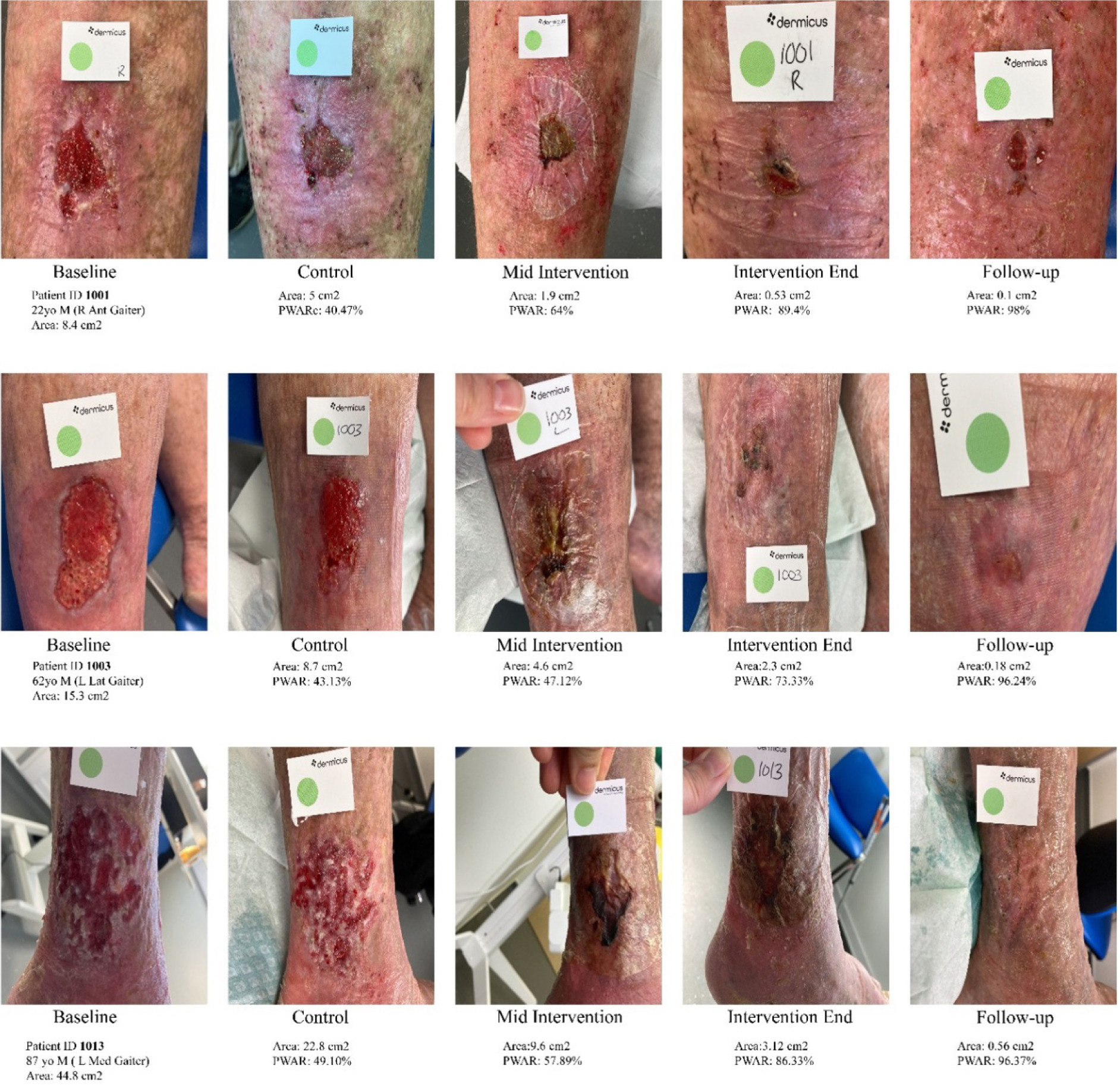
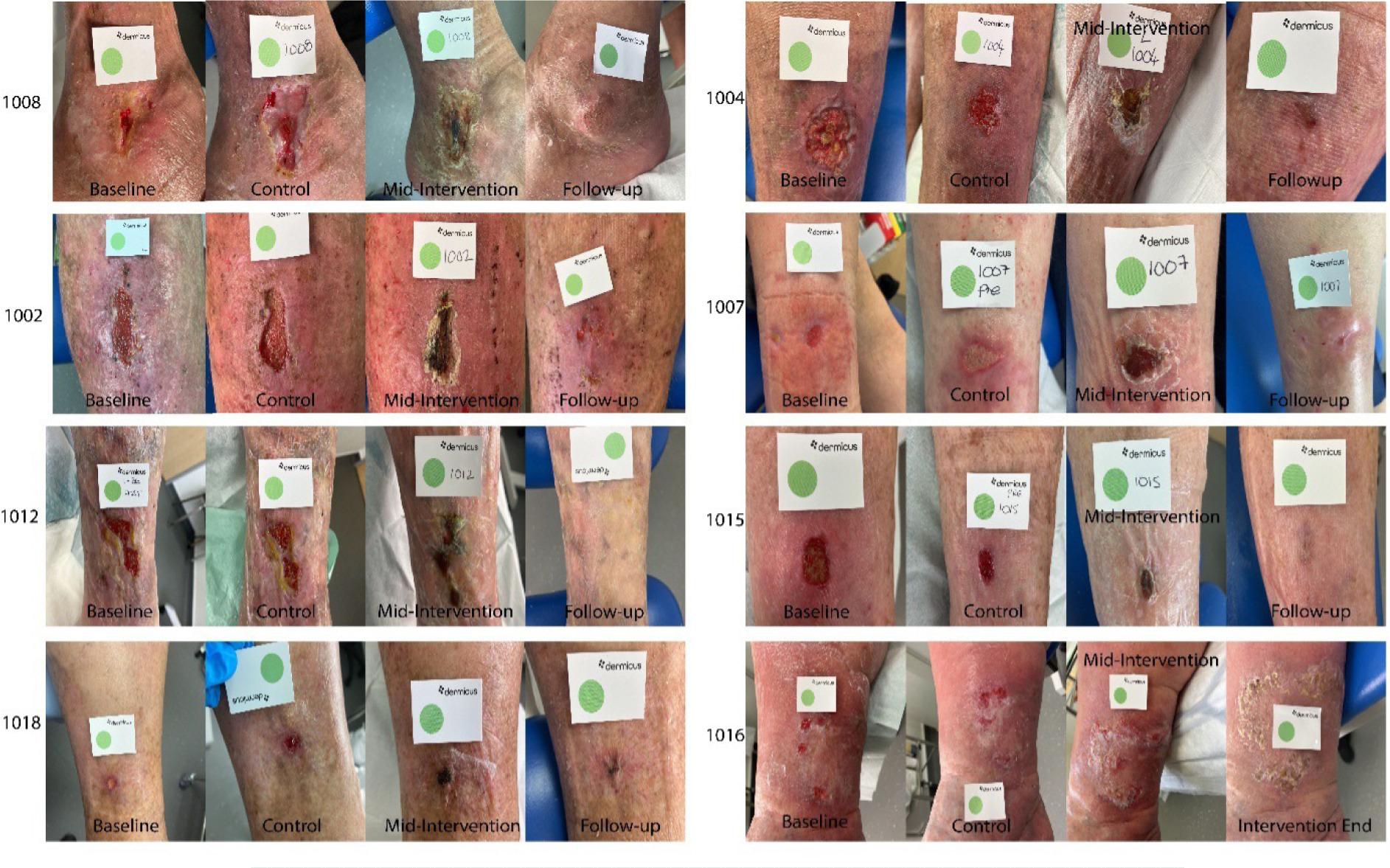
FIGURE 6 Representative examples of wound progression among Epiprotect-treated participants. Sequential wound photographs (Baseline → Control → Mid-intervention → Follow-up) from eight participants (IDs 1002, 1004, 1007, 1008, 1012, 1015, 1016, 1018) demonstrate consistent contraction, granulation, and epithelialisation following transition to Epiprotect. Control-phase images typically show static or inflamed wound beds, while mid- and late-intervention images reveal reduced exudate, cleaner granulating tissue, and re-epithelialised margins. All wounds were chronic venous leg ulcers imaged using Dermicus under standardised lighting and orientation. Progressive healing is visible across all cases, with substantial improvement by the 12-week follow-up.
Discussion
Interpretation of findings
This service evaluation demonstrated that Epiprotect, a BC dressing, was associated with substantial and progressive improvements in wound healing among adults with chronic VLUs treated in routine community practice. PWAR improved following the transition from standard care to Epiprotect, and sustained improvements were noticed at follow-up. Most participants achieved meaningful healing thresholds (≥75% or ≥90%), and three achieved complete closure. These findings suggest that BC dressings may be associated with improved healing trajectories compared with the preceding standard-care phase, although causal inference beyond temporal association is not possible in this study design.
The rapid reduction in pain after Epiprotect application is consistent with previous BC research, where patients frequently report early analgesic effects. In this cohort, pain scores fell to zero by weeks 10-12, which aligns with studies in burns and pediatric wounds showing reduced nociceptive stimulation and improved comfort. Patients and nursing staff in this evaluation also reported enhanced comfort and reduced time burden, reinforcing the practical advantages of a dressing that adheres securely and requires minimal reapplication.
Significant reductions in exudate further support Epiprotect's capacity to stabilize the wound microenvironment. Excess exudate is a well-recognized barrier to healing in VLUs, contributing to maceration, inflammation, and impaired keratinocyte migration. The structured reduction observed in this study suggests that Epiprotect may contribute to improved moisture balance, consistent with BC's high water-binding capacity and breathable nanofibrillar matrix.
Role of dressing adherence in healing
A key finding is the significant correlation between early adhesion of Epiprotect and subsequent healing outcomes. Better adherence by day 3 was associated with higher PWAR and smaller final wound sizes and exudate levels. In contrast, participants needing more dressing sheets, often because of early detachment caused by heavy exudate, showed slower healing progress.
The mechanism underlying Epiprotect's adherence to the wound bed remains incompletely understood. The manufacturers acknowledge that the precise biochemical interactions have not been fully elucidated. A plausible hypothesis is that its hydroxyl-rich, nanofibrillar network may promote protein–polymer interactions at the tissue interface, facilitating stable adhesion and moisture retention. However, direct mechanistic evidence is lacking, and this represents a significant gap in the translational science of BC materials. The consistent association between adhesion and healing in this study reinforces the need for further mechanistic research, potentially incorporating histological analysis, surface chemistry studies, and in situ imaging of the BC–tissue interface.
Importantly, even in cases where full adherence was not initially achieved, improvements in granulation quality and peri-ulcer skin condition were consistently observed, suggesting that Epiprotect may exert additional benefits beyond adherence alone.
The initial application required perfection, with the wound debrided and no air bubbles trapped between the sheets and the wound bed upon application of Epiprotect. This was followed by a wait for the Epiprotect to adhere before completing the compression dressing. However, good adherence led to several benefits in subsequent visits, including reduced dressing time, improved wound visibility, and fewer dressing changes due to better exudate control. Patient-reported comfort and pain relief were also noted, with one patient expressing a desire to continue using the dressing beyond the study.
Comparison with existing literature
The results of this study largely align with existing research on BC dressings for chronic VLUs. Multiple studies have documented pain relief, effective exudate control, and good patient tolerance, findings that closely match our own.41 For example, Cavalcanti et al42 observed notable improvements in peri-ulcer skin condition and patient comfort using BC membranes. However, they did not find a statistically significant reduction in wound size over a 120-day period.42 Likewise, Dini et al43 found higher epithelialization rates with BC dressings than with saline gauze, but no significant difference in total healing time. Pain reduction was a consistent theme across all studies, including ours. The average dressing use of 2.2±1.25 per participant further indicates prolonged wear and high compliance during the intervention, aligning with prior research that observed similar relief with Eiratex, with minimal dressing changes, averaging 1.7 per patient. Patients in this evaluation reported a mean VAS score of 0 by week 12, consistent with prior research, such as that by Sivlér et al.28
Notably, despite the expanding literature on BC dressings, a lack of comparative trials directly evaluating Epiprotect against other advanced wound dressings, including alternative BC products, foams, hydrofibers, or multilayer systems, remains. Current evidence largely derives from burns care or small pilot series, many of which lack standardized wound-healing metrics or structured follow-up. This limits the ability to contextualize Epiprotect's performance within the broader spectrum of advanced wound care technologies. The present study highlights the need for controlled, head-to-head comparisons to determine whether Epiprotect offers unique clinical advantages or cost benefits compared to other established wound therapies.
Limitations and future work
The self-controlled, non-randomized design limits causal inference and does not fully account for time-dependent healing, regression to the mean, or increased clinical attention following intervention, all of which may have contributed to observed improvements. The pilot study's self-controlled design risks biases from timing, behavior, and natural healing, as it lacks randomization or blinding, which can affect outcomes such as pain and adherence. Patients were aware that they were using a new dressing, which could have introduced expectation bias. Wound-healing differences may reflect natural progression despite standard care, according to NICE guidelines. Selection bias could occur due to differences in wound sizes and first-come, first-served inclusion criteria across the two centers. The small final sample size (n=12) limits generalizability and power, increasing the risk of type II errors.
This study employed a non-validated, clinician-derived exudate scoring scale based on PUSH and BWAT principles,44,45 demonstrating internal consistency but lacking formal validation. Future research should include validated measures and patient-reported outcome tools, such as Wound-QoL or EQ-5D, to assess clinical and quality-of-life outcomes.
The study's small size, limited number of centers, and self-controlled design restrict its broader relevance. Using photographic wound measurements, even with ImageJ calibration, might miss deeper tissue or long-term changes. Future research should include randomized, blinded, adequately powered trials with longer follow-up periods.
To verify these initial findings, a multicenter randomized controlled trial (RCT) is needed with patient and assessor blinding, a larger and more diverse sample, and stratification by ulcer size and depth (small, medium, large). Including biological assessments, such as markers for granulation, inflammation, and epithelialization, would clarify the mechanisms. Further RCTs should compare Epiprotect with standard multilayer treatments and other biosynthetic dressings to assess effectiveness. Longer follow-up and validated patient-reported outcomes, like quality of life and cost-effectiveness, would enhance future research.
Conclusion
This study assessed the real-world use of a BC dressing for chronic VLU management and examined its associations with healing and pain outcomes. Results indicate that BC dressings may be associated with better healing outcomes than the previous standard-of-care phase, although the study design does not allow for causal conclusions beyond temporal correlation.
This study provides a credible foundation for early data, paving the way for more comprehensive research. These promising findings support the development of a multicenter randomized controlled trial comparing Epiprotect with standard care and other BC dressings. Future studies should include a diverse population with stratified ulcer depth, ulcer sizes, biological markers, histopathology, and long-term outcomes. Overall, this service evaluation represents a crucial initial step toward integrating this novel BC dressing into evidence-based chronic wound management.