Introduction
Despite advances in wound healing technologies, recurrence rates in diabetic lower extremity wounds remain high, with major amputations often considered inevitable once chronic ulceration or osteomyelitis is established.Conventional approaches focus on wound closure and infection control but rarely address the biomechanical drivers of ulcer recurrence.
The VISTA strategy—Vascular Evaluation, Infection Control, Surgical Correction, Technologies & Therapies, Amputation Prevention—emerges as a comprehensive solution. VISTA integrates advanced wound care with proactive surgical correction and long-term patient commitment to prevent limb loss. It reframes surgical intervention not as a last resort, but as a foundational and concurrent element of diabetic wound care.
Methods
Strategy development
The VISTA model was derived from an evidence-based synthesis of current literature on DFUs, surgical deformity correction, vascular interventions, infection control, and wound technologies.
Literature review
Databases searched included PubMed, MEDLINE, and Google Scholar. Keywords included: “diabetic foot ulcer”, “wound recurrence”, “vascular assessment”, “foot deformity correction”, “Charcot reconstruction”, “osteomyelitis”, “bone infection”, “bioactive biomaterials”, and “limb salvage”. Inclusion criteria focused on studies addressing surgical outcomes, recurrence rates, and integrated wound care strategies in high-risk diabetic populations.
The VISTA strategy
V — Vascular evaluation
Perfusion is foundational. Without adequate blood flow, neither surgical nor conservative therapies can succeed.1 Vascular status should be assessed at the first point of specialist contact in all diabetic foot wounds, even in the absence of overt ischemic signs.
Key strategies include:
-
Non-invasive vascular testing:
-
Ankle-brachial index (ABI): Though commonly used, may be falsely elevated in diabetic patients due to medial arterial calcification.1,2
-
Toe-brachial index (TBI): More reliable in diabetics for assessing distal perfusion.
-
Near-infrared spectroscopy (NIRS): This modality, including the plantar-palmar index method, provides a noninvasive option for evaluating microvascular perfusion and peripheral arterial disease, particularly when ABI or TBI values are unreliable in diabetic populations. Recent studies have validated NIRS as a reliable alternative for assessing tissue perfusion in diabetic limbs and the plantar-palmar index with near infrared spectroscopy as an alternative to the ankle-brachial index.2
-
Skin perfusion pressure (SPP) and transcutaneous oxygen measurement (TcPO2): Provide direct assessment of microcirculation and wound healing potential.1,3
-
Early referral for formal vascular consultation (surgery or interventional radiology/cardiology) is critical when peripheral arterial disease is suspected/identified.1,4
-
-
Imaging modalities:
-
Duplex ultrasonography: First-line imaging for identifying stenoses or occlusions.
-
Computed tomography angiography (CTA) or magnetic resonance angiography (MRA): provide detailed vascular mapping when intervention is considered.
-
-
Revascularization planning:1,3,4
-
Early referral to vascular surgery or interventional radiology is critical when perfusion deficits are identified.1,4
-
Endovascular interventions (angioplasty, stenting) and open bypass surgeries should be tailored based on anatomy, patient comorbidities, and wound severity.1,3
-
Revascularization should precede major reconstructive or debridement procedures to optimize healing potential.1,3
-
-
Multidisciplinary coordination:
I — Infection control
DFUs are frequently complicated by deep infections, including osteomyelitis. Infection is not merely a side effect of chronic wounds but often a driving factor in progression to amputation.6-8 VISTA emphasizes early, aggressive infection management, including both systemic and localized strategies—combined with surgical source control.
Key principles:
-
Prompt diagnosis and stratification: Clinical signs of infection must be corroborated with lab tests (C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), white blood cell (WBC)), imaging (MRI/CT), and bone biopsy when osteomyelitis is suspected.6,8,9
-
Culture-guided antibiotic therapy: Empiric antibiotics are used initially, but definitive treatment must be tailored to culture and sensitivity.8,9
-
Surgical source control: Includes sharp debridement, drainage of abscesses, removal of necrotic tissue, and resection of infected dead bone.8,9
-
Minimally invasive interventions:
-
Bone–percutaneous limb antibiotic salvage treatment (B-PLAST): A dual-port system for percutaneous delivery of antibiotic-eluting bone void fillers (e.g., Cerament-G), performed under fluoroscopic guidance. B-PLAST allows targeted infection management while preserving foot architecture. It can treat infected viable bones. If an infected bone has a blood supply and structural integrity, it can most likely be salvaged.7,8,10 The technique can be re-utilized in the same bone.
-
-
Advanced imaging guidance:
-
Fluorescence-guided bacterial visualization aids in identifying bacterial load beyond what is visible clinically, optimizing debridement precision. Several available bacterial autofluorescence technologies have shown efficacy in identifying wound bacterial location and load (>104) as well as guiding more effective bedside debridement.11
-
-
Multidisciplinary approach: Infectious disease specialists collaborate with surgeons and wound clinicians to align antimicrobial therapy with surgical timing and healing phases.
-
Adjunctive local therapies:
Infection control in VISTA is not delayed or secondary—it is integrated into the initial assessment and used to determine the wound’s potential for tissue salvage, timing of closure, and eligibility for structural correction.7-9
S — Surgical correction
Surgical correction is pivotal to breaking the cycle of recurrent diabetic foot wounds by addressing the underlying biomechanical and structural abnormalities that predispose to ulceration. VISTA elevates surgical intervention from a last-resort measure to an integral, early component of comprehensive diabetic foot management.7,13
Core principles
-
Timely intervention: Early identification and correction of deformities—before the onset of severe ulceration or infection—can prevent progression to limb-threatening complications.7,13
-
Comprehensive deformity correction: Correction of bony and soft tissue abnormalities reduces pathological plantar pressures and abnormal joint biomechanics, thereby lowering recurrence risk.7,13
-
Multimodal surgical approaches: VISTA supports both minimally invasive and open surgical techniques tailored to individual patient pathology and comorbidities.5,7
Key surgical techniques
-
Tendon and soft tissue procedures:
-
Bony procedures:
-
Osteotomies and arthrodesis (fusion) procedures for correcting midfoot and hindfoot deformities, such as Charcot neuroarthropathy or midfoot collapse.5,7
-
Lapidus bunionectomy for first ray instability and hallux valgus deformities.7
-
Metatarsal head resection or shortening to relieve localized plantar pressure zones.7,13
-
-
External fixation:
-
Soft tissue reconstruction:
Surgical staging and integration
-
Vascular assessment and needed intervention combined with infection control is prioritized prior to definitive reconstruction.
-
Procedures are often staged, beginning with debridement and infection clearance, followed by structural correction and then soft tissue reconstruction.
-
Early involvement of multidisciplinary teams ensures coordinated perioperative management and glycemic control.
-
Simultaneous bone and soft tissue reconstruction when indicated, minimizing surgical stages and optimizing functional outcomes.
Outcomes and evidence:
-
Studies have demonstrated that surgical correction of deformities reduces ulcer recurrence rates significantly compared to conservative care alone.7,13
-
Limb salvage rates improve with proactive deformity correction, particularly in patients with Charcot foot and recurrent ulcers.7
-
Minimally invasive approaches reduce surgical morbidity and support faster recovery when appropriate.5,7
T — Technologies and therapies
VISTA harnesses a dynamic and evolving clinical armamentarium of advanced tools, devices, and therapeutic modalities designed to optimize wound healing, support structural preservation, and precisely target infection and biomechanical abnormalities. These innovations enable minimally invasive interventions and personalized care, elevating treatment efficacy while reducing morbidity.7,11,12
-
Negative pressure wound therapy with instillation (NPWTi) and antimicrobial dressings enhance wound bed preparation and infection control.8,12
-
Antibiotic-eluting bone void fillers deliver localized antibiotics while preserving bone architecture in osteomyelitis management.8,10
-
Smart external fixation systems provide multiplanar deformity correction combined with real-time biomechanical monitoring.5,7
-
Advanced diagnostics including thermal imaging, near-infrared spectroscopy (NIRS), and BAF-guided bacterial visualization improve assessment of tissue perfusion and infection burden.2,11
-
Pressure-mapping technologies, custom 3D-printed orthotics, and wearable sensors support biomechanical offloading and patient adherence.7,11,13
By leveraging these state-of-the-art technologies and therapies, clinicians can deliver targeted, effective, and personalized interventions that promote durable healing and functional limb salvage.
A — Amputation prevention
Amputation prevention within VISTA represents a comprehensive, patient-centered clinical pathway focused on long-term preservation of limb function, quality of life, and avoidance of limb loss. This approach transcends acute wound care to incorporate durable offloading, rehabilitative strategies, patient education, and vigilant surveillance. 7,8,14
-
Durable offloading strategies including custom therapeutic footwear and orthoses minimize plantar pressures and protect vulnerable tissues.7,13
-
Structured gait retraining and physical therapy restore functional mobility and redistribute mechanical load.7
-
Routine clinical follow-up augmented by advanced imaging detects early signs of ischemia, re-ulceration, or biomechanical compromise, enabling timely, preemptive interventions.1,2,7
-
Patient education empowers self-care, adherence to offloading, and systemic risk factor control such as glycemic management.7,14
-
Multidisciplinary collaboration ensures integrated management across vascular surgery, infectious disease, endocrinology, podiatry, and rehabilitation disciplines.1,4,5
Embedded within this longitudinal framework, amputation prevention shifts the clinical focus from reactive salvage to proactive, sustained limb preservation—thereby improving patient outcomes and reducing healthcare costs related to major limb loss.7,14
Case 1: Staged midfoot reconstruction for chronic lateral fifth metatarsal ulceration
Patient Profile
A 61-year-old male with a medical history of type 2 diabetes mellitus (A1c 7.4%), hypertension, hyperlipidemia, chronic obstructive pulmonary disease (COPD), arthritis, and prior pneumonia presented with a chronic ulcer over the lateral aspect of the left fifth metatarsal. Past surgical history included appendectomy, coronary artery bypass grafting, cardiac catheterization, bilateral cataract extraction, right hemicolectomy, and total knee arthroplasty.
VISTA framework integration
V – Vascular evaluation
Non-invasive vascular testing with ankle-brachial index demonstrated mild small vessel disease at the transmetatarsal level bilaterally. There was no evidence of critical limb ischemia, and perfusion was deemed adequate to support surgical healing. No revascularization was indicated.
I – Infection control
Initial imaging revealed osteomyelitis of the left fifth metatarsal. The patient was hospitalized and started on intravenous antibiotics. An incision and drainage was performed with debridement of infected tissue and placement of antibiotic-eluting bone void filler as a local antibiotic carrier. Deep tissue cultures guided ongoing antibiotic therapy. The preoperative appearance of the chronic ulcer over the lateral fifth metatarsal is shown in Figure 1.
FIGURE 1 Preoperative photograph of chronic ulcer over the lateral left fifth metatarsal
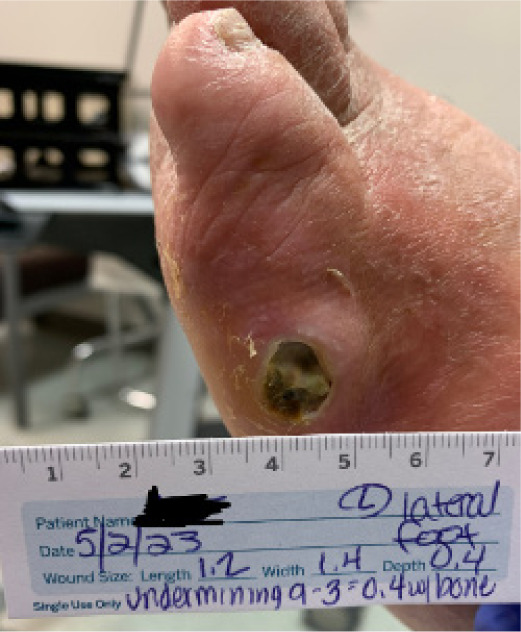
S – Surgical correction
Two days after the initial debridement, the patient underwent corrective surgery to address the underlying structural deformity. Procedures included correction of metatarsus adductus, Lapidus bunionectomy, and application of an external fixator to stabilize the midfoot and offload the lateral column (Figure 2).
FIGURE 2 Postoperative photograph showing external fixator placement with midfoot realignment.
At follow-up 3 months later, the patient presented with a new abscess at the lateral foot. He was taken back to surgery for repeat incision and drainage with curettage of the fifth metatarsal and repeat application of antibiotic-eluting bone void filler. Negative pressure wound therapy (NPWT) was applied at this stage to promote granulation and manage drainage.
T – Technologies and therapies
Antibiotic bone void filler was used in both operative settings to provide localized antibiotic delivery to the infected fifth metatarsal. NPWT was implemented after the second debridement to optimize the wound bed and support closure.
A – Amputation prevention
The combination of staged debridement, structural realignment, external fixation, and localized antibiotic delivery prevented progression to more proximal amputation. The patient was transitioned to custom diabetic footwear and orthotics following healing to maintain correction and offload high-pressure zones. Glycemic control continued to be an emphasis with education.
Outcome
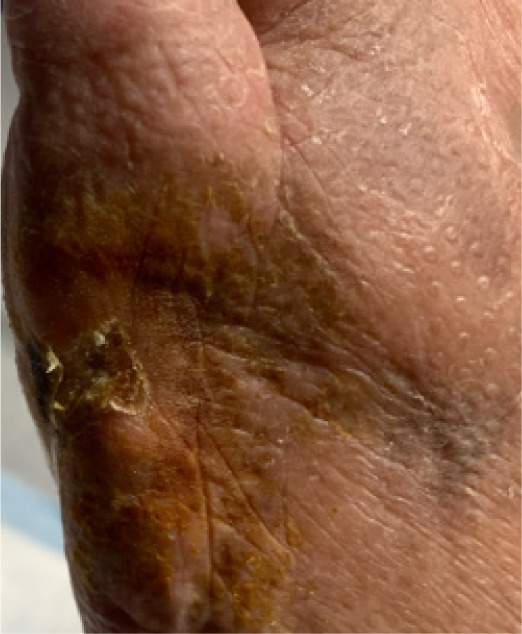
The ulcer healed fully by mid-2024 following staged reconstruction, infection control, and prolonged IV antibiotic therapy. The lateral left foot remained ulcer-free with stable alignment and no recurrence at latest follow-up (Figure 3).
FIGURE 3 Preoperative photograph of chronic ulcer over the lateral left fifth metatarsal.
Case 2: Staged Charcot reconstruction and osteomyelitis management using external fixation and local antibiotic delivery
Patient profile
A 56-year-old male with a history of type 2 diabetes mellitus (HbA1c 10.5%), peripheral neuropathy, prior femoropopliteal bypass surgery, and former tobacco use presented with a chronic plantar midfoot ulcer. The wound was associated with underlying osteomyelitis and deformity consistent with midfoot Charcot neuroarthropathy. Radiographic and clinical findings demonstrated rocker-bottom collapse with bony prominence at the ulcer site and instability in the midfoot.
VISTA framework integration
V – Vascular evaluation
Non-invasive vascular testing including toe-brachial index confirmed adequate microvascular perfusion. Duplex ultrasonography demonstrated patency of the prior femoropopliteal bypass graft. No additional revascularization procedures were necessary prior to surgical intervention. The patient was cleared for limb-salvage surgery without delay.
I – Infection control
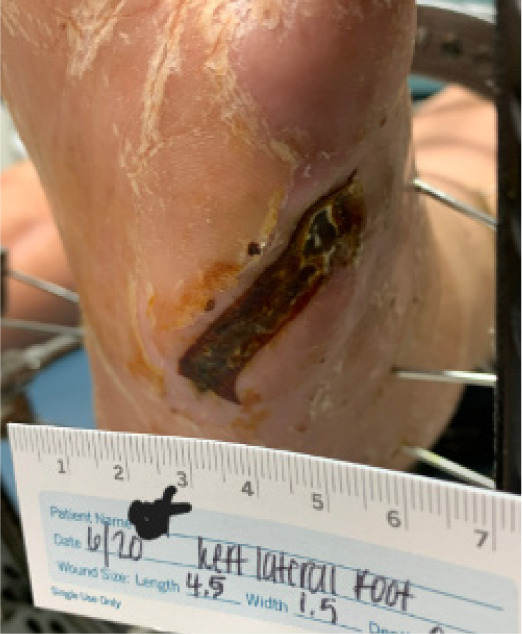
The chronic ulcer probed to bone and was confirmed as osteomyelitis by MRI and positive intraoperative cultures (methicillin-sensitive Staphylococcus aureus and coagulase-negative staphylococci) (Figure 4). Laboratory workup revealed elevated inflammatory markers (ESR and CRP), consistent with chronic deep infection. Surgical debridement was performed with resection of necrotic bone and application of a bioabsorbable, antibiotic-impregnated bone void filler. Infectious disease consultation guided culture-specific intravenous antibiotics, and a multi-week antimicrobial regimen was coordinated around the surgical timeline.
FIGURE 4 Preoperative image showing plantar ulceration over collapsed midfoot.
S – Surgical correction
A three-stage surgical approach was performed to address both infection and mechanical instability. Stage one involved midfoot debridement with application of antibiotic-impregnated bone void filler for local antibiotic delivery and dead space management (Figure 5). In stage two, a multiplanar external fixator was applied to achieve gradual realignment of the midfoot and provide stable offloading of the ulcerated plantar surface (Figure 6). Finally, stage three included adjunctive procedures such as hammertoe correction, plantar exostectomy, and ankle arthrodesis to address associated instability and deformities contributing to the patient’s altered gait mechanics.
FIGURE 5 Intraoperative view showing antibiotic-impregnated bone void filler placement in debrided midfoot void.
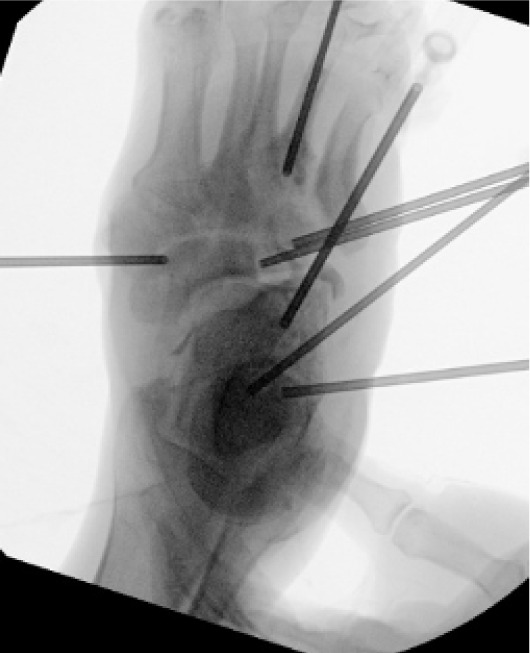
FIGURE 6 External fixator applied for stabilization and realignment. Graft present at wound site.
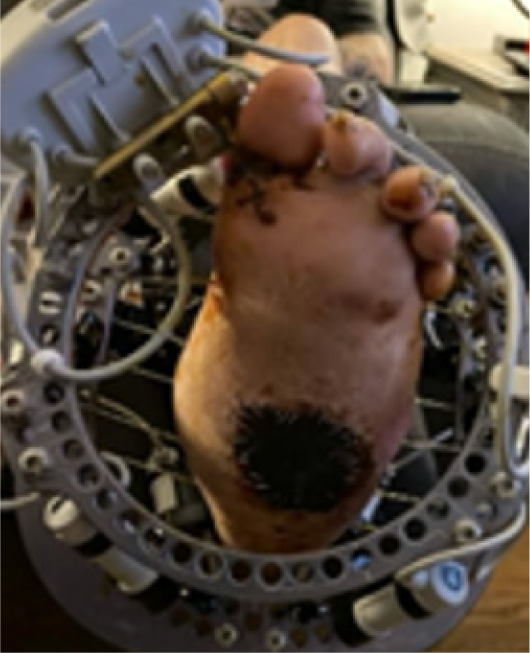
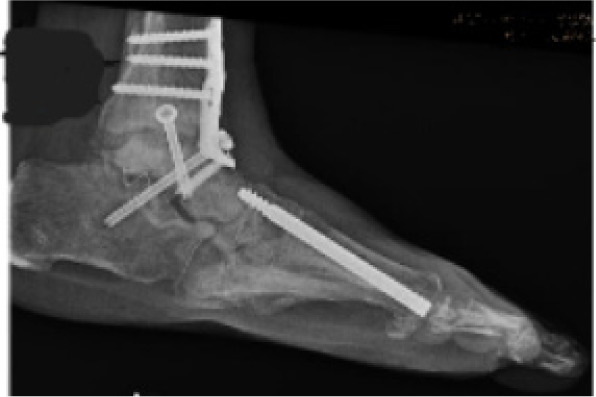
FIGURE 7 Postoperative radiograph after frame removal showing stable fusion and corrected midfoot alignment.
T – Technologies and therapies
Advanced wound technologies were used throughout the staged process. Antibiotic-impregnated bone void filler provided dual function as an antibiotic delivery system and bone void filler. A multiplanar circular dynamic external fixator facilitated controlled alignment correction while maintaining wound offloading. Negative pressure wound therapy was used during initial phases to optimize granulation tissue and manage exudate. Antibiotic beads impregnated with gentamycin were used intraoperatively to provide local antibiotic delivery. Postoperative radiographs confirmed progressive consolidation and maintenance of alignment
A – Amputation prevention
The comprehensive surgical plan successfully preserved the midfoot and avoided below-knee amputation. After frame removal, the patient was transitioned to custom offloading Charcot-resistant footwear and enrolled in an ongoing diabetic foot prevention program. Glycemic optimization was addressed in partnership with endocrinology, and biomechanical surveillance was maintained through podiatric follow-up.
Outcome
The patient achieved full ulcer healing following external frame removal. At 9 months postoperatively, a minor recurrence was noted at the prior ulcer site, which was successfully treated with localized debridement and a second application of antibiotic-impregnated bone void filler without requiring new fixation. At the 14-month follow-up, the patient remained ambulatory in diabetic footwear, without ulcer recurrence or progression to major amputation (Figure 8). Functional outcome was excellent, and the patient reported no limitations in daily mobility.
FIGURE 8 Healed image of previous plantar ulcer.
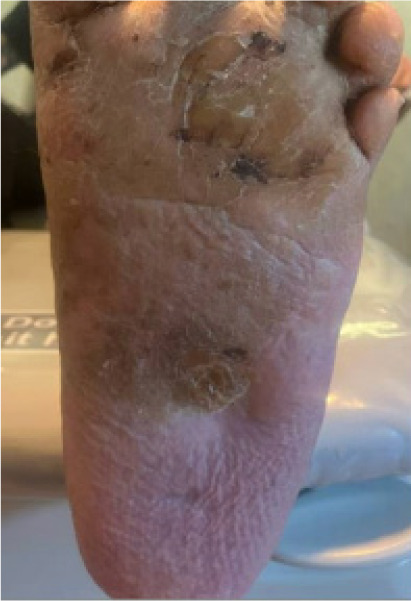
Case 3: multistage reconstruction for chronic lateral forefoot ulceration and forefoot deformities in a diabetic patient
Patient profile
A 47-year-old male with a history of type 2 diabetes mellitus, hypertension, peripheral neuropathy, and anxiety presented with a chronic tunneling ulcer located at the lateral aspect of the right forefoot. The ulcer had persisted despite offloading and standard wound care. Clinical examination revealed rigid hammertoe deformities of the lateral digits and a significant hallux valgus deformity contributing to medial instability and lateral overload. The patient had previously failed conservative management, and imaging demonstrated cortical irregularity of the lateral metatarsal consistent with chronic osteomyelitis.
VISTA framework integration
V – vascular evaluation
Initial vascular workup included an ankle-brachial index (ABI) and venous duplex ultrasound, The ABI indicated adequate perfusion, and the venous duplex showed no evidence of significant venous reflux. Although ABI alone can be falsely elevated in diabetic patients due to medial arterial calcification, the clinical exam supported sufficient perfusion for surgical healing. No signs of limb-threatening ischemia were present, and the patient was cleared for surgical intervention without need for further vascular imaging or revascularization.
I – Infection control
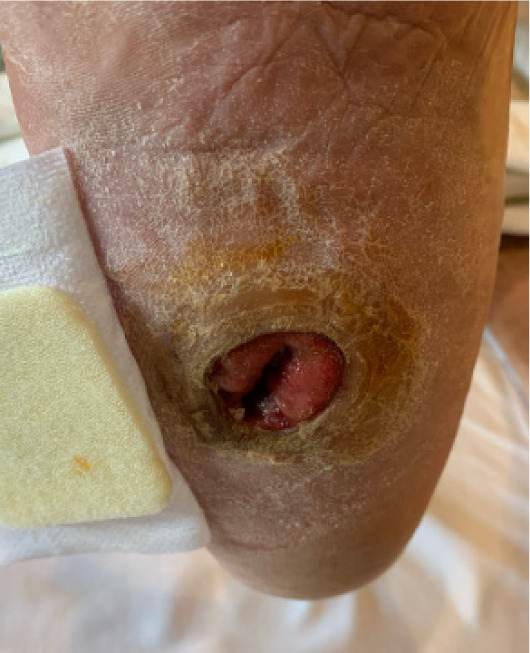
The tunneling ulcer probed to bone, and MRI confirmed localized osteomyelitis involving the fifth metatarsal head (Figure 9). Bone cultures were obtained intraoperatively, and surgical curettage of the infected bone was performed. Culture-guided antibiotic therapy was initiated in collaboration with infectious disease. Local management included weekly debridement and moist wound healing with antimicrobial dressings during the initial phase.
FIGURE 9 Wound image showing lateral forefoot ulcer with surrounding callus and digit deformities.
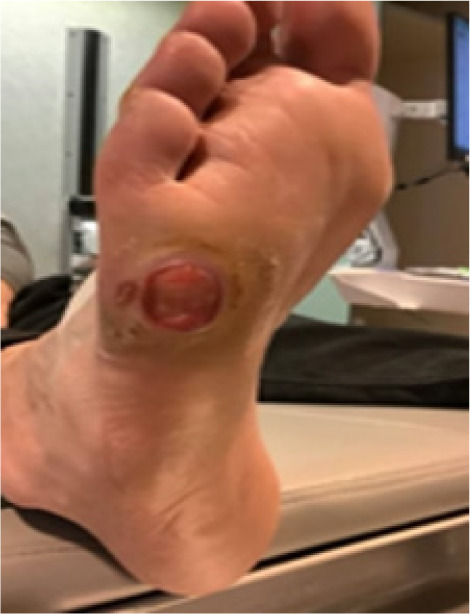
S – Surgical correction
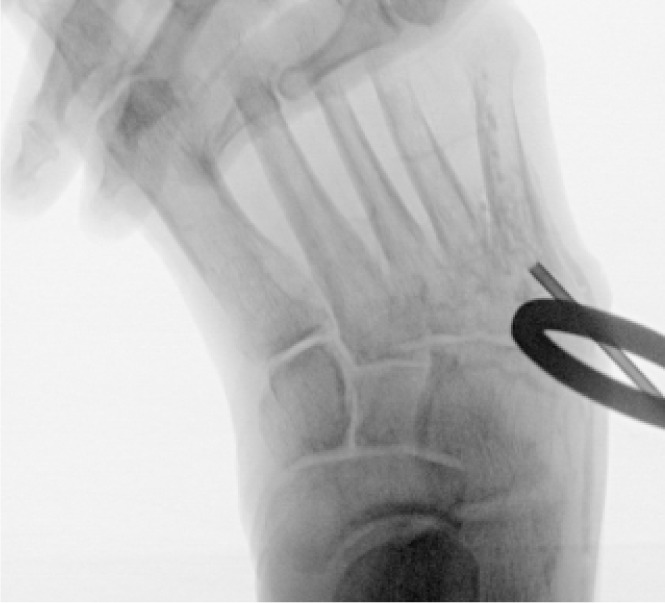
A staged surgical approach was implemented to address both the infectious and structural components contributing to the chronic ulcer. In the first stage, the patient underwent curettage and limited resection of the infected fifth metatarsal head. After thorough debridement, a bioabsorbable antibiotic-sulfate bone void filler was placed into the osseous defect to promote local antimicrobial activity and reduce dead space (Figure 10). This procedure was performed in conjunction with aggressive offloading and wound care to support granulation and early epithelialization.
FIGURE 10 Intraoperative image showing placement of antibiotic-impregnated bone void filler into the lateral metatarsal defect.
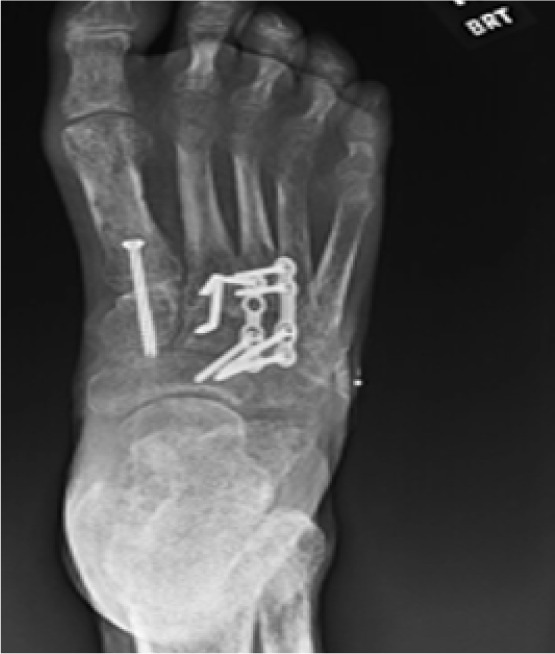
Following clinical resolution of infection, the second stage involved definitive deformity correction. A Lapidus bunionectomy with fusion of the first tarsometatarsal joint was performed to correct medial column instability and hallux valgus deformity. Concurrently, hammertoe corrections of the second through fifth digits were completed using arthrodesis techniques to address rigid digital contractures contributing to abnormal lateral forefoot pressure (Figure 11).
FIGURE 11 Postoperative image at 12 weeks showing healed lateral forefoot with corrected alignment.
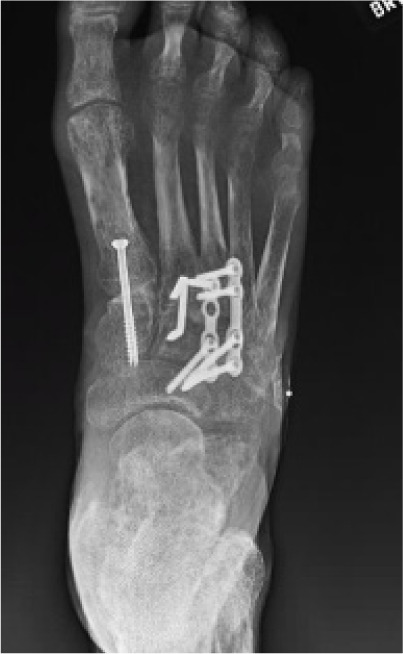
In the third and final stage, elective removal of hardware was performed after radiographic evidence confirmed successful fusion and alignment (Figure 12). This was done to reduce long-term risk of hardware prominence or skin breakdown, particularly given the patient’s history of ulceration and peripheral neuropathy.
FIGURE 12 Radiograph confirming Lapidus fusion consolidation and digital realignment.
T – Technologies and therapies
Antibiotic-impregnated bone void filler was used during the initial surgical debridement to both eradicate local infection and fill cortical voids. Custom postoperative offloading devices were employed throughout the surgical recovery phases. Targeted wound care included collagen dressings, antimicrobial copper therapies, and silicone-based foam padding. Postoperatively, the patient was enrolled in structured physical therapy and gait retraining to address longstanding compensatory mechanics and restore balanced ambulation.
A – Amputation prevention
Amputation was avoided through early surgical source control, timely deformity correction, and integration of advanced wound technologies. The patient was transitioned into custom footwear with forefoot offloading and pressure mapping-guided orthotic adjustment. Ongoing podiatric and endocrine follow-up was maintained to monitor for recurrence, and the patient was educated on daily foot care and glycemic control as key components of long-term limb preservation.
Outcome
The tunneling ulcer resolved by 6-month follow up, following staged reconstruction and infection control. At 10-month follow-up, the patient remained ulcer-free and fully weight-bearing in custom diabetic footwear. Foot alignment was preserved, and the patient reported substantial improvement in balance and mobility. No new areas of pressure or ulceration were noted. Preventative surveillance remains ongoing with no need for further surgical intervention to date.
Discussion
Recurrent wounds require structural solutions
DFU recurrence remains 40–65% within 5 years.7 Pressure redistribution strategies alone are insufficient without addressing the mechanical drivers—hammertoes, Charcot collapse, tight heel cords. VISTA’s emphasis on early deformity correction breaks this cycle.
Innovation enables minimally invasive, targeted care
Technologies such as antibiotic-eluting bone fillers, smart external fixation, and biologic grafts allow effective infection control and healing while preserving structural integrity. VISTA integrates these tools to support proactive care even in high-risk or frail patients.
Multidisciplinary care enhances outcomes and efficiency
Coordinated input from vascular surgeons, infectious disease specialists, endocrinologists, podiatry, cardiology, orthopedists, and wound care providers is essential. VISTA provides a shared roadmap for these teams, reducing fragmentation and improving outcomes.
Cost-effectiveness and systemic impact
Preventing even a single major amputation reduces costs associated with prolonged hospitalization, prosthetics, and rehabilitation.14 Early surgical correction and structured pathways lower the burden on both patient and system.5,7
Conclusion
VISTA delivers an unequivocal, forward-thinking paradigm shift in diabetic limb preservation by seamlessly integrating rigorous early vascular assessment, uncompromising infection control, precise and timely surgical correction, alongside cutting-edge innovative therapies. This dynamic, limb-centric approach decisively moves beyond the limitations of reactive wound management, driving toward sustainable healing and robust functional limb salvage. By harnessing the VISTA framework—which prioritizes proactive detection and management of vascular insufficiency, infection, and biomechanical deformity—clinicians are empowered to foresee and pre-empt complications with precision, substantially elevating patient outcomes while dramatically reducing amputation incidence. The framework’s promise underscores the urgent need for comprehensive multicenter trials to rigorously establish its transformative impact on long-term limb preservation and healthcare system sustainability.