Introduction
Pressure injuries (PIs), also referred to as pressure ulcers, decubitus ulcers, or bedsores, are localized injuries to the epidermis and/or underlying tissue that occur most often over bony prominences due to sustained pressure and shear, frequently compounded by moisture, microclimate, and impaired perfusion. They are common in frail and immobilized populations and are associated with prolonged care needs, infection risk, hospital utilization, and mortality.1,2
Stage 3 PIs represent a clinically consequential threshold because they involve full-thickness skin loss with penetration into the adipose tissue layer; granulation tissue, slough/eschar and undermining/tunneling may be present. By definition, deeper structures such as fascia, muscle, tendon, or bone are not exposed or directly palpable. Stage 3 wounds are frequently resource-intensive and may progress to systemic complications (e.g., sepsis) if not effectively treated and offloaded.1
The burden of PIs is rising with aging demographics. A 2025 Global Burden of Disease analysis focused on adults ≥60 years of age found global incident cases increased from 1990 to 2021, with projections suggesting further increases in several countries, including the United States.3 In the U.S., inpatient trend analyses have suggested that overall prevalence may remain relatively stable across some periods while severity increases.4 Beyond human impact, PIs generate substantial economic burden; modeling studies of hospital-acquired PIs estimate national costs in excess of $27 billion annually, with a disproportionate share attributable to advanced-stage wounds.5 U.S. policy and quality programs have targeted hospital-acquired PIs for more than a decade, highlighting persistent system-level incentives to prevent advanced-stage complications and downstream hospital-based resource utilization, including emergency department visits, inpatient admissions, and intensive care services.6,7
Recent international consensus guidance on cellular, acellular, and matrix-like products (CAMPs) in PI ulcers highlights that the PI ulcer evidence base remains substantially less developed than for other hard-to-heal wound etiologies, such as the multitude of real-world evidence studies on CAMPs to effectively treat lower extremity diabetic ulcers (LEDUs) and venous leg ulcers (VLUs).8-10 Thus, the 2025 international consensus guidance, “Cellular, Acellular and Matrix-Like Products (CAMPs) in Pressure Injuries” cautions against extrapolating outcomes from studies of CAMPs used in other wound etiologies to the treatment of PIs.11
The same consensus document calls for complementary real-world evidence to address gaps in generalizability and to evaluate outcomes aligned with patient-centered goals in medically fragile populations, including reduction in infection, hospitalization, and mortality.11 In parallel, a multi-stakeholder U.S. consensus on CAMP regulatory and reimbursement reform emphasizes strengthening patient access and fiscal stewardship in coverage and payment policy and specifically recommends that future coverage frameworks incorporate real-world evidence and expand coverage beyond LEDUs/VLUs to include other hard-to-heal wounds, such as PIs.12
In this context, advanced wound therapies, including CAMPs, are increasingly used as adjuncts to standard wound care. However, real-world effectiveness and downstream utilization outcomes in high-risk Medicare populations with Stage 3 PIs remain incompletely characterized. Accordingly, defining the real-world clinical and utilization impact of CAMP use in Stage 3 PIs in a national Medicare population is important for informing evidence-based care pathways and policy decisions for a high-risk, high-cost condition. This study evaluates Stage 3 PI outcomes among Medicare beneficiaries using national claims data, comparing episodes treated with CAMPs plus standard of care (SOC) versus SOC alone.
Methods
This retrospective cohort study evaluated Medicare beneficiaries with Stage 3 PIs using the CMS 100% Limited Data Set (LDS), including Inpatient, Outpatient, and Master Beneficiary Summary File claims from 2016–2024. The LDS is de-identified; the study was considered exempt from IRB oversight.
Episode construction and exclusions
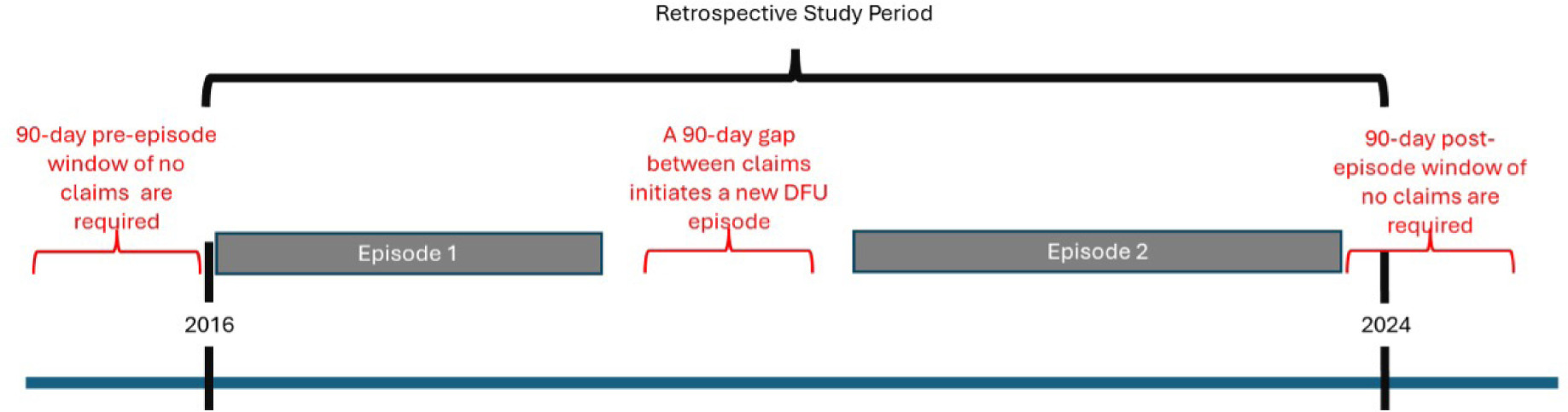
Treatment episodes were defined as continuous wound-related care periods separated by ≥90-day clean period (defined as ≥90 days without wound-related services) (Figure 1). To ensure complete boundary capture, episodes that began within the first 90 days or ended within the final 90 days of the observation window were excluded. Episodes were removed if beneficiaries had end stage renal disease or invalidated if no medical spending occurred during the episode as the absence of reimbursed claims suggested that no billable wound-related treatment or standard care was captured for comparative analysis. Ulcer location (including sacral and heel) was captured and incorporated into matching.
Treatment groups
Two groups were analyzed:
-
SOC group: episodes of care that included sharp debridement and no CAMP.
-
CAMP group: episodes receiving SOC plus a CAMP billed under Q- or A- HCPCS codes as detailed in previous work.13
Because treatment episodes in administrative claims frequently include sequential or multiple CAMP products and because HCPCS coding may represent product categories rather than specific products, product-level comparative effectiveness analyses were not performed. Accordingly, this study evaluated the impact of CAMP therapy at the category level, comparing episodes in which any CAMP was incorporated into care with episodes treated with SOC alone. This approach reflects the structure of claims data and the real-world clinical practice pattern in which CAMP products may be selected or changed over the course of wound management.
Comorbidity and clinical risk adjustment
Comorbidity burden was summarized using the Charlson Comorbidity Index (CCI) derived from diagnosis codes during the episode and the preceding 90 days. Severe protein-calorie malnutrition and Class II/III obesity (BMI ≥35 kg/m2) were included to improve baseline comparability.
FIGURE 1 Study design and episode definition. The retrospective study period initiated on October 1, 2016 and continued to Oct 2, 2024. Each episode (grey bars) was evaluated using claims data. Episodes were assigned as new if no claims data existed for 90 days prior and continued until resolved or another 90-day gap in claims occurred. A 90-day gap following a claim was also required for an episode to be considered resolved.
Matching
A 1:1 matching strategy paired CAMP and SOC episodes using exact-match and range-based variables. Exact matches included gender, sacral location, heel location, debridement ≥20 cm2, severe protein-calorie malnutrition, and BMI ≥35 kg/m2; range matching included age category, days to treatment initiation, CCI category, and episode start year loosely based on pre-COVID, during COVID and post-COVID eras (2016–2019, 2020–2021, 2022–2024). Analyses were performed in IBM SPSS.
Outcomes and statistics
Outcomes included treatment timing (days to first intervention, episode length), clinical outcomes (major/minor amputation, infection-related complications, mortality defined as death within 90 days of episode end), utilization (ED visits, readmissions, intensive care unit/critical care unit (ICU/CCU) encounters, hospitalizations), and costs (total all-cause and PI-diagnosis–associated spending). Continuous variables used independent-samples t-tests and categorical variables used chi-square tests (p<0.05). Analytic subset: Stage 3 episodes were evaluated in a 100% Hospital Outpatient Department (HOPD)/inpatient sample with episode minimums (90 and 120 days) comparing CAMPs + SOC versus SOC without CAMPs. Two minimum-duration cohorts (≥90 and ≥120 days) were analyzed as a sensitivity analysis to ensure that findings were robust to episode-definition choices and to preferentially capture sustained, clinically meaningful Stage 3 PI care trajectories.
Results
Patient demographics and treatments
A total of 110,381 PI episodes (99,517 treated with SOC, and 10,864 that received a CAMP) were eligible for study matching. PI locations were primarily sacral or the heel (Figure 2). The average Medicare enrollee with a PI was a Caucasian around 71-year-old male (Table 1). They had an average CCI score of 4, with more than 30% of enrollees having at least one of the following comorbidities: complicated diabetes, peripheral vascular disease, congestive heart failure, renal disease, chronic obstructive pulmonary disease or paralysis (Table 2). Comorbidities and wound-care procedures for each minimum cohort are summarized in Table 2.
In this study all subjects received debridement, as that was required for a wound measurement. Other adjunctive treatments were used for a minority of patients. Only 26.8% of episodes that were over 120-days and received a CAMP were provided with negative pressure wound therapy (NPWT) which dropped to 13.6% for episodes of 90-120 days without CAMP treatment. Most other adjunctive treatments were provided to less than 10% of episodes (Table 2).
Cohort size and baseline comparability
-
Episode minimum 90 days (Stage 3, HOPD-only): 7,223 matched episodes per group (CAMP vs SOC).
-
Episode minimum 120 days (Stage 3, HOPD-only): 6,139 matched episodes per group.
Across both minimum-duration cohorts, matched groups were similar in age, sex, and CCI, with no meaningful differences post-match.
Complications of PIs range from quality of life (QoL) issues to death. Propensity matched cohorts were analyzed for the frequency of mortality, sepsis and major amputations (Figure 3). Nearly 40% of enrollees receiving SOC developed sepsis, and 22% of all enrollees in the SOC cohort died, underscoring the high complication burden in this population.
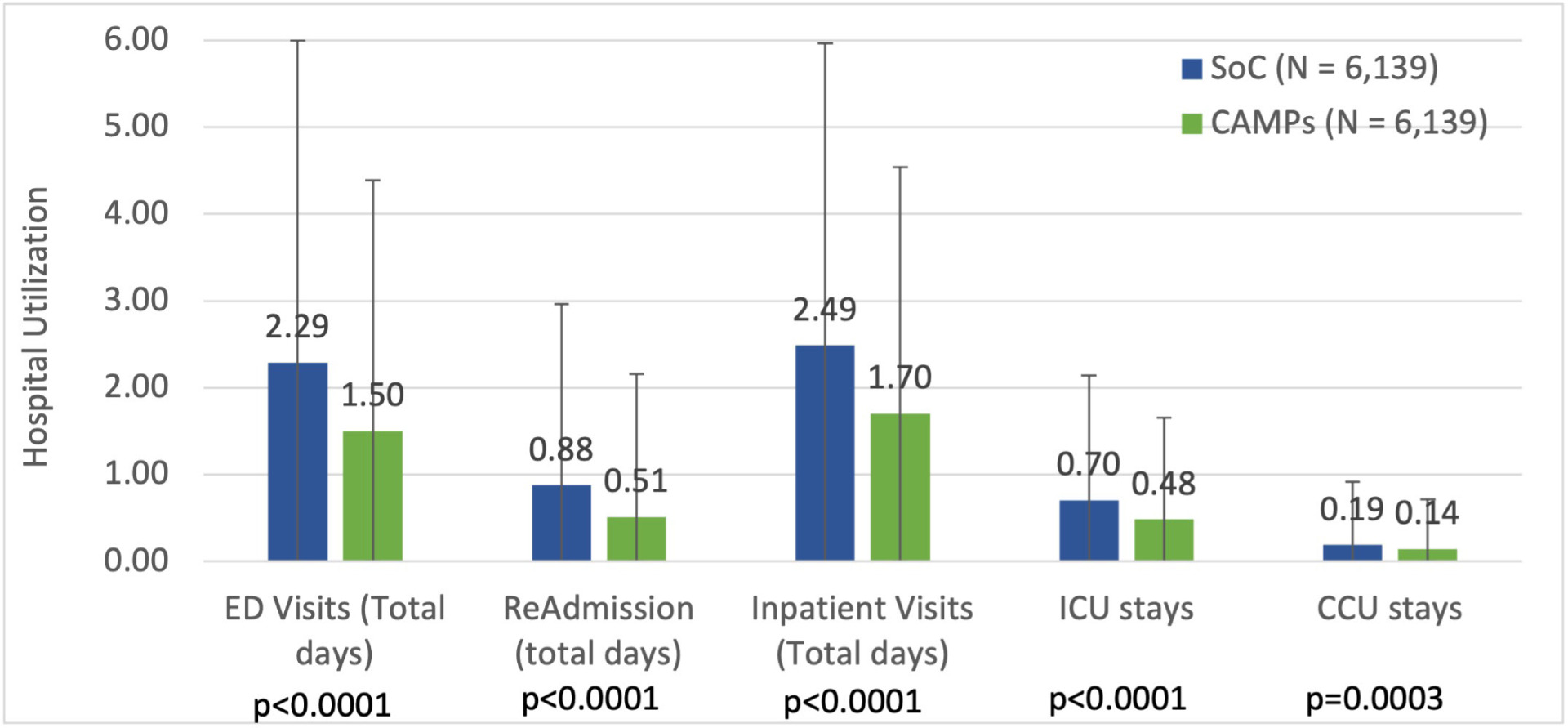
In all cases Medicare enrollees treated with a CAMP had statistically lower complication rates than those who received SOC only. The effectiveness of treatment was evaluated by hospital-based acute-care utilization during the episode, including ED visits, readmissions, inpatient visits, and ICU/CCU. Visits to the ED averaged over 2.2 days per episode for those receiving SOC and 2.4 days of inpatient stays. Additionally, hospital readmissions, ICU, and cardiac care unit (CCU) stays were compared between cohorts and statistically significant reductions were observed with CAMP treatment (Figure 4).
FIGURE 2 Pressure injury ulcer locations of 99,517 Medicare episodes that received standard care.
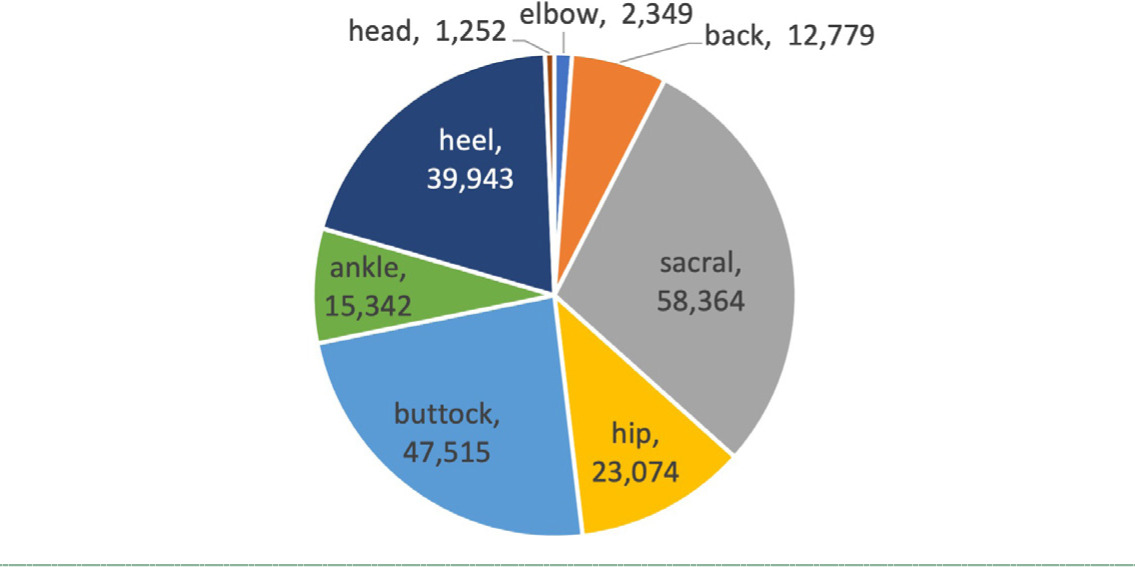
TABLE 1 Demographics
| Stage 3 Min 90 days | Stage 3 Min 120 days | |||||
|---|---|---|---|---|---|---|
| SOC N=7,223 |
CAMPs N=7,223 |
P value | SOC N=6,139 |
CAMPs N=6,139 |
P value | |
| Age | 70.9 | 71.0 | 0.88 | 70.0 | 70.2 | 0.56 |
| Gender (M) | 53.4% | 53.4% | 1.00 | 53.5% | 53.5% | 1.00 |
| Race (W) | 81.4% | 86.0% | P<0.001 | 80.9% | 85.6% | P<0.001 |
| Race (B) | 13.2% | 8.2% | P<0.001 | 13.5% | 8.6% | P<0.001 |
| Race (H) | 1.8% | 1.9% | 0.68 | 1.8% | 2.0% | 0.34 |
| Race (O) | 3.6% | 3.9% | 0.31 | 3.8% | 3.7% | 0.79 |
| Dual eligible Medicare + Medicaid | 37.92% | 30.89% | P<0.001 | 40.38% | 32.22% | P<0.001 |
| Medicare Part A | 99.10% | 99.31% | 0.09 | 99.01% | 99.35% | 0.01 |
| Medicare Part B | 99.86% | 99.97% | 0.08 | 99.87% | 99.97% | 0.08 |
| Medicare Part C | 3.99% | 3.54% | 0.15 | 4.01% | 3.52% | 0.14 |
| CCI score | 3.99 | 4.00 | 0.77 | 4.10 | 4.10 | 0.97 |
M, male; W, white;B, black; H, hispanic; O, other; CCI, Charlson Comorbidity Index
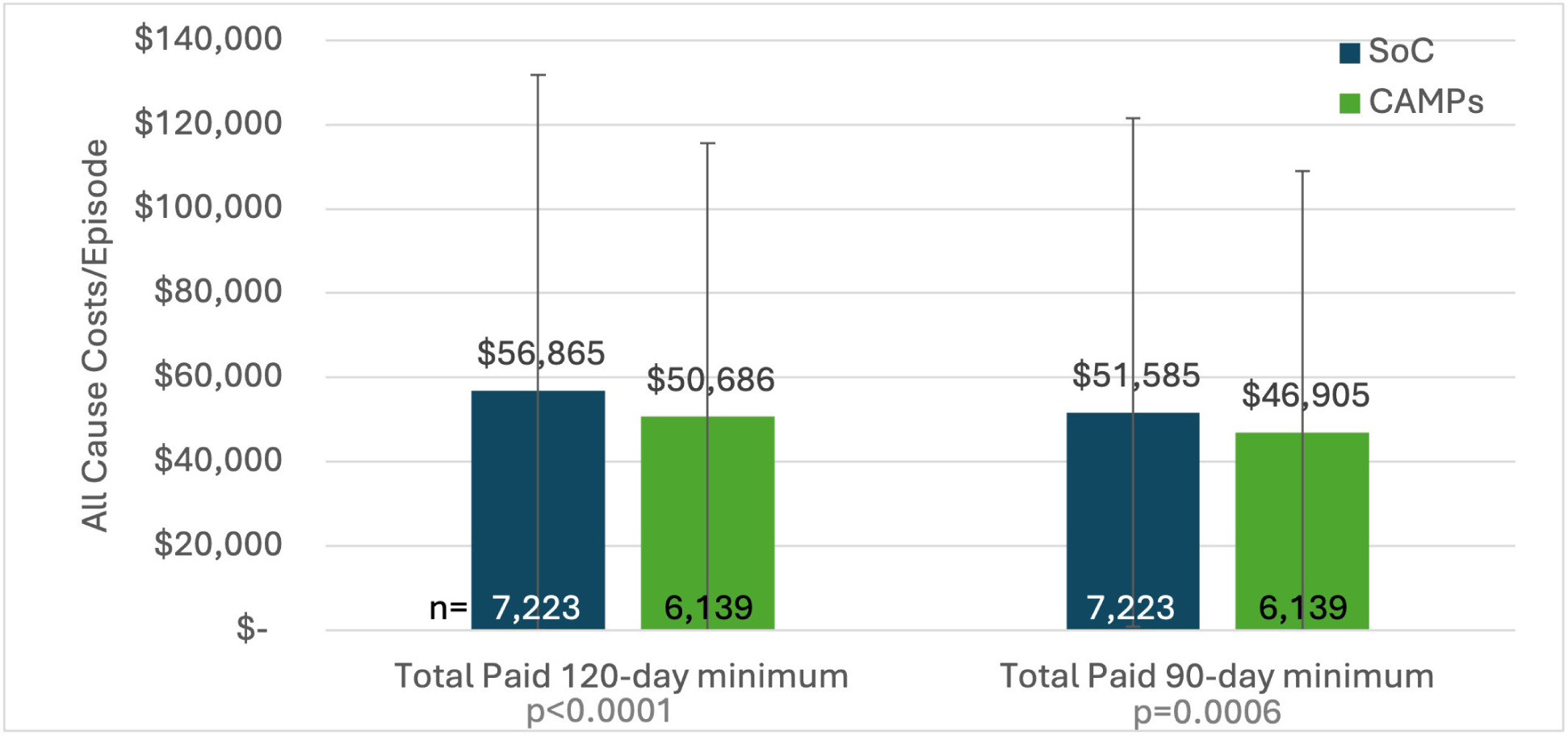
On average, all cause claim costs were $46,905 to $56,865. Costs were tabulated for each cohort and reductions of $6,179 and $4,680 were identified for 120 and 90-day minimum episodes when a CAMP was used (Figure 5). Interpretation of directionality:
-
CAMP episodes showed lower acute-care utilization (ED, inpatient, ICU/CCU), lower sepsis, lower major amputation, and lower mortality across both episode-duration thresholds.
-
CAMP episodes had longer time-to-first intervention and longer episode length, yet were associated with lower overall and PI-coded spending.
PI location patterns (overall Stage 3 sample descriptors)
Across recorded locations, the most common Stage 3 PI sites in the dataset included sacral, buttock, and heel regions, consistent with typical pressure-exposed bony prominences (Figure 2).
Discussion
In this national Medicare claims analysis (2016–2024) focused on Stage 3 PIs, CAMP-treated episodes were associated with meaningfully lower downstream health-care utilization, lower sepsis, and lower mortality, alongside lower total and PI-related spending. These findings align with the clinical premise that CAMPs, when used as an adjunct to foundational care (debridement, moisture balance, offloading, and risk-factor optimization), may help reduce complications that drive hospital-based costs and adverse outcomes.
The observed reductions in sepsis, acute-care utilization, and mortality among CAMP-treated episodes are directionally consistent with the international PI consensus, which emphasizes that clinically meaningful success in this population extends beyond wound closure to broader patient-centered outcomes such as infection prevention, hospitalization avoidance, and survival.6 The consensus further underscores that effective use of CAMPs depends on foundational elements, such as patient optimization (e.g., nutrition and comorbidity management), pressure redistribution/offloading, and wound bed preparation, which may plausibly contribute to reduced downstream complications that drive inpatient escalation.6
By establishing a well-balanced analytical foundation, the interpretation of these findings should account for the biological and systemic determinants that govern wound-healing trajectories. Hard-to-heal wounds arise within a complex host context, and their progression is often driven less by diagnostic categorization than by the extent to which systemic and local barriers (e.g., impaired perfusion, chronic inflammation/infection, malnutrition, and inadequate pressure redistribution) disrupt the universal phases of tissue repair.12 These considerations are especially relevant in real-world Medicare populations, where multimorbidity and frailty may overshadow differences attributable to any single advanced therapy12 and may influence both the timing of CAMP initiation and downstream utilization outcomes.13 We note that the average Medicare enrollee who developed a PI had a CCI score of about 4.
TABLE 2 Comorbidities and care procedures of analyzed Medicare patients with pressure injuries*
| 90-day minimum episode | 120-day minimum episode | |||||
|---|---|---|---|---|---|---|
| SOC (N=7,223) |
CAMPs (N=7,223) |
SOC (N=6,139) |
CAMPs (N=6,139) |
P value | ||
| Comorbidities | Mean | Mean | P value | Mean | Mean | P value |
| Diabetes mellitus complicated | 38.80% | 42.20% | p<0.001 | 39.20% | 42.10% | 0.002 |
| Peripheral vascular disease | 37.50% | 44.10% | p<0.001 | 37.80% | 44.60% | p<0.001 |
| Congestive heart failure | 34.20% | 33.70% | 0.55 | 33.60% | 34.00% | 0.65 |
| Renal disease | 31.10% | 32.20% | 0.15 | 31.80% | 32.50% | 0.4 |
| Chronic obstructive pulmonary disease | 30.30% | 29.30% | 0.19 | 31.50% | 30.20% | 0.11 |
| Paralysis | 29.70% | 26.60% | p<0.001 | 32.60% | 29.00% | p<0.001 |
| Dementia | 26.20% | 18.40% | p<0.001 | 25.90% | 18.70% | p<0.001 |
| Abnormalities of gait and mobility | 20.50% | 17.50% | p<0.001 | 21.00% | 18.30% | p<0.001 |
| Severe protein-calorie malnutrition | 10.40% | 10.40% | 1 | 11.60% | 11.60% | 1 |
| Cardiovascular disease | 17.10% | 15.30% | 0.004 | 17.70% | 16.20% | 0.03 |
| Unspecified protein-calorie malnutrition | 14.10% | 9.80% | p<0.001 | 15.30% | 10.70% | p<0.001 |
| BMI 35.0 or greater (35.0–45.9 range) | 8.20% | 8.20% | 1 | 8.50% | 8.50% | 1 |
| Myocardial infarction | 15.20% | 15.70% | 0.39 | 15.40% | 16.10% | 0.27 |
| BMI >35 (36.0–45.9 range) | 7.50% | 7.60% | 0.84 | 7.80% | 7.80% | 1 |
| Moderate protein-calorie malnutrition | 12.20% | 9.20% | p<0.001 | 13.30% | 10.10% | p<0.001 |
| BMI 19.9 or less, adult | 9.70% | 7.90% | p<0.001 | 10.70% | 8.80% | 0.001 |
| Cancer | 10.70% | 11.40% | 0.19 | 10.90% | 11.50% | 0.3 |
| Adult failure to thrive | 7.70% | 4.60% | p<0.001 | 9.50% | 5.00% | p<0.001 |
| Other reduced mobility | 8.50% | 6.10% | p<0.001 | 8.60% | 6.70% | p<0.001 |
| Functional quadriplegia | 6.20% | 4.20% | p<0.001 | 7.40% | 4.70% | p<0.001 |
| Rheumatic | 7.10% | 8.60% | 0.001 | 6.80% | 8.80% | p<0.001 |
| Peptic ulcer disease (PUD) | 3.70% | 3.30% | 0.22 | 3.90% | 3.60% | 0.36 |
| Need for assistance with personal care | 3.20% | 2.00% | p<0.001 | 3.80% | 2.20% | p<0.001 |
| Age-related physical debility | 3.60% | 2.50% | p<0.001 | 3.50% | 2.70% | 0.01 |
| Mild protein-calorie malnutrition | 3.50% | 2.50% | 0.001 | 3.80% | 2.70% | 0.001 |
| Liver mild | 2.00% | 1.70% | 0.15 | 1.90% | 1.90% | 1 |
| Immobility syndrome | 1.40% | 1.60% | 0.29 | 1.60% | 1.80% | 0.42 |
| Liver severe | 1.60% | 1.40% | 0.27 | 1.90% | 1.50% | 0.08 |
| Stiffness of unspecified joint | 0.50% | 0.20% | 0.02 | 0.40% | 0.30% | 0.36 |
| HIV | 0.10% | 0.20% | 0.23 | 0.10% | 0.30% | 0.04 |
| Paralytic gait | 0.10% | 0.10% | 1 | 0.10% | 0.10% | 1 |
| Wound care procedures | Mean | Mean | P value | Mean | Mean | P value |
| Debridement | 100.0% | 100.0% | — | 100.0% | 100.0% | — |
| Negative pressure wound therapy | 13.60% | 24.50% | p<0.001 | 14.80% | 26.80% | p<0.001 |
| Debridement (>20 cm2) - muscle | 6.50% | 7.50% | 0.02 | 7.10% | 8.30% | 0.02 |
| High compression bandage | 6.10% | 12.50% | p<0.001 | 6.60% | 12.20% | p<0.001 |
| Debridement (>20 cm2) - bone | 2.30% | 2.50% | 0.45 | 2.60% | 2.70% | 0.78 |
| Hyperbaric oxygen treatment | 1.40% | 4.10% | p<0.001 | 1.50% | 4.40% | p<0.001 |
| Total contact cast | 0.60% | 2.30% | p<0.001 | 0.80% | 2.30% | p<0.001 |
| Venous ablation | 0.10% | 0.10% | 1 | 0.00% | 0.10% | 0.04 |
| Compression stocking | 0.10% | 0.10% | 1 | 0.00% | 0.10% | 0.04 |
*All PIs were Stage 3 from only the hospital outpatient setting.
TABLE 3 Clinical outcomes by treatment group and episode duration threshold (Stage 3 PI episodes; ≥90 days vs ≥120 days)
| Outcome | 90d SOC | 90d CAMP | p-value | 120d SOC | 120d CAMP | p-value |
|---|---|---|---|---|---|---|
| Days to first intervention | 119.78 | 126.45 | 0.0017 | 131.93 | 139.23 | 0.0031 |
| Episode length (days) | 302.13 | 323.77 | <0.0001 | 339.08 | 359.61 | <0.0001 |
| PI-diagnosis spending | 39,886 | 36,397 | 0.0003 | 45,266 | 40,075 | <0.0001 |
| Total all-cause spending | 50,686 | 46,905 | 0.0006 | 56,865 | 51,585 | <0.0001 |
| Major amputation | 3.10% | 2.20% | 0.0004 | 3.30% | 2.40% | 0.0041 |
| Sepsis | 36.10% | 26.10% | <0.0001 | 39.90% | 28.80% | <0.0001 |
| Mortality | 22.30% | 17.80% | <0.0001 | 22.80% | 18.40% | <0.0001 |
| ED visits (during episode)/1000 | 2,044.0 | 1,339.6 | <0.0001 | 2,288.8 | 1,501.7 | <0.0001 |
| Readmissions/1000 | 762.1 | 454.7 | <0.0001 | 876.9 | 513.4 | <0.0001 |
| Inpatient visits/1000 | 2,235.9 | 1,523.6 | <0.0001 | 2,486.9 | 1,703.4 | <0.0001 |
| ICU utilization/1000 | 627.9 | 436.8 | <0.0001 | 704.5 | 484.0 | <0.0001 |
| CCU utilization/1000 | 179.0 | 129.3 | <0.0001 | 186.7 | 143.3 | 0.0003 |
d, days; ED, emergency department; ICU, intensive care unit; CCU, cardiac care unit.
Against this backdrop, utilization and outcome differences should be interpreted in the context of real-world treatment sequencing and episode construction. Notably, CAMP-treated episodes had a longer delay to first application and longer overall episode duration, with more outpatient encounters. In practice, CAMPs are often introduced after initial stabilization, diagnostic workup (including perfusion and infection assessment), wound bed preparation, and failure of early standard measures, so claims-based “episode length” may reflect delayed initiation rather than prolonged non-healing. In addition, longer observed episodes may represent continued outpatient surveillance and maintenance care intended to prevent relapse, manage undermining/tunneling and bioburden, and avoid inpatient escalation. Importantly, Stage 3 PIs involve full-thickness tissue loss and are prone to undermining/tunneling and contamination; preventing progression to deep infection, sepsis, and hospitalization is central to improving outcomes.14,15
FIGURE 3 Major complications for Medicare enrollees with Stage 3 PIs who received standard of care (SOC, blue bars) propensity matched to those who received a CAMP (green bars). Subjects were all from the HOPD setting with a minimum episode length of 120-days.
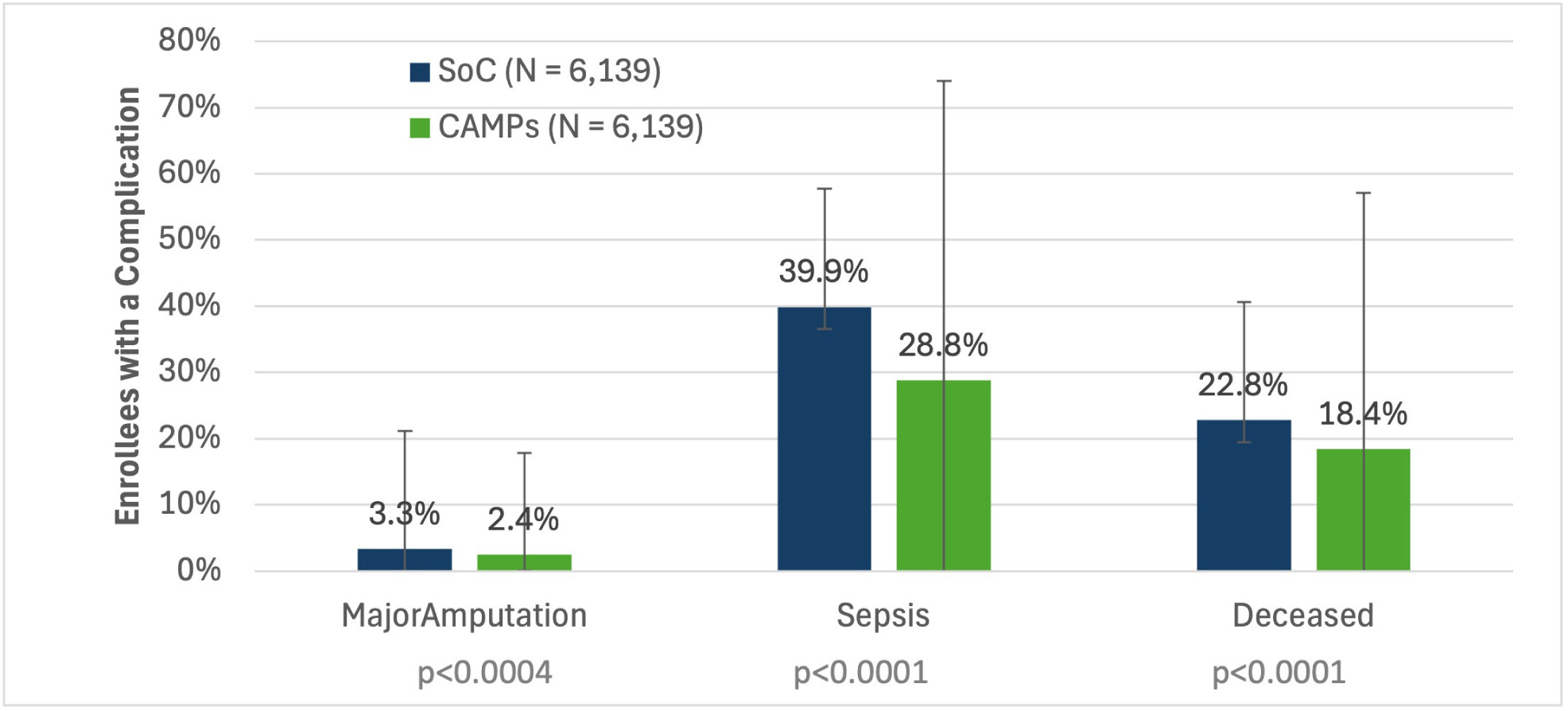
FIGURE 4 Hospital-based acute-care utilization for Medicare enrollees with Stage 3 PIs who received standard of care (SOC, blue bars) propensity matched to those who received a CAMP (green bars). Subjects were all from the HOPD setting with a minimum episode length of 120-days.
Consistent with this high-risk clinical phenotype, Medicare enrollees with Stage 3 PIs experienced high rates of severe complications (Table 3, Figure 3). Nearly 40% of those receiving SOC developed sepsis and 22% of all enrollees under SOC alone died. These are depressing outcomes. While it is positive to see significant improvements with the use of CAMPs, other adjunctive treatments appear to be underutilized. No more than 26% received NPWT in the cohorts examined here. Venous ablation or HBO2 therapy were listed on less than 5% of claims (Table 2). It seems intuitive that more frequent usage of established treatments will improve outcomes.
When care for patients with PIs is lacking, one would expect poor outcomes, frequent recurrence and many additional hospital visits. Indeed, claims for enrollees under SOC alone saw high hospital utilization rates, suggesting that enroll-ees are not getting sufficient treatment. On average, SOC alone enrollees are spending 4.6 days of total time combined in the ED and inpatient settings. Such usage leads to high expenses and further impacts availability of hospital resources. When we examined all claim costs during episodes of 120-days or more, a total of $56,865 was tallied for those under SOC alone. When we examined only claims where a PI was the primary diagnosis, costs dropped by about $10,000; nevertheless, SOC with CAMPs was associated with about $5,000 lower total episode costs than SOC alone. This suggests that appropriate use of CAMPs can yield meaningful savings.
FIGURE 5 All cause claim costs for Medicare enrollees with Stage 3 PIs who received SOC (blue bars) propensity matched to those who received a CAMP (green bars). Subjects were from the HOPD setting. Comparison of episodes with a minimum length of 90-days are compared to 120-days. Size of each cohort is labeled at the base of each bar..
Health economic and coverage policy implications
Beyond direct episode costs, U.S. inpatient trend analyses suggest that PI case-mix severity may be increasing over time, even when prevalence appears stable, making the high inpatient-origin share of PI episodes particularly consequential amid contracting inpatient resources and tightening bed capacity. Using CMS provider of services files, acute-care inpatient facility counts declined by ≈7% from 2018 to 2025, and certified bed counts declined by ≈2% over the same period. In parallel, nearly half of PI episodes meeting the minimum-duration thresholds began with an inpatient admission. Taken together, these patterns suggest that policy or coverage changes that shift PI care toward hospital-based settings may further stress inpatient capacity and increase competition for beds needed for higher-acuity medical and surgical admissions. Accordingly, preserving feasible outpatient access to effective advanced wound therapies is not only a patient-centered priority but also a health-system capacity strategy.
As this analysis focuses on HOPD episodes, it provides a relevant analogue to CMS payment approaches that constrain CAMP reimbursement through packaged/bundled or fixed-fee mechanisms. The “Regulatory and Reimbursement Reform for Cellular, Acellular, and Matrix-like Products (CAMPs)” consensus, which included Dr. David G. Armstrong as one of the panelists, cautions that limiting CMS’ analysis to 2024 HOPD-only 4th quarter claims inputs may underestimate the broader post-acute/non-facility care mix and recommends that rate-setting incorporate volume-weighted claims across all sites of care. It further asserts that any tiered payment approach should be driven by demonstrable differences in clinical outcomes rather than regulatory categorization alone.12 Accordingly, these findings inform expectations for future CAMP coverage policy in facility settings and warrant parallel evaluation in non-facility settings. From a Medicare perspective, the observed reductions in severe complications and hospital-based utilization are clinically and economically meaningful. Across both episode-duration cohorts, episodes treated with CAMPs were associated with lower rates of sepsis and major amputation and fewer ED visits, inpatient admissions, and ICU/CCU encounters. These improvements occurred despite longer episodes and more outpatient visits, suggesting a shift in care away from costly inpatient escalation toward sustained outpatient management.
The consistency of cost findings across episode definitions strengthens the economic signal. In both the 90-day and 120-day minimum cohorts, CAMP-treated episodes had lower PI-related spending and lower total all-cause spending. Because these estimates reflect allowed amounts in Medicare claims, they capture the combined effects of wound care services, facility encounters, and complication-related utilization. Importantly, greater outpatient intensity and longer episode duration did not translate into higher overall spending when downstream complications were reduced. Taken together, these utilization and cost patterns warrant consideration of their potential implications at the Medicare program level.
Budget impact and relevance to national coverage policy
To contextualize the observed differences in utilization and costs, we conducted a simple claims-based budget impact assessment using per-episode spending observed in the matched cohorts. The intent was to estimate potential Medicare program implications associated with broader CAMP use in a defined Stage 3 PI ulcer population, rather than to perform a formal cost-effectiveness analysis.
In the 90-day minimum cohort, mean total all-cause spending was $3,781 lower per episode with CAMPs compared with standard care ($46,905 vs $50,686). In the 120-day minimum cohort, the corresponding difference was $5,280 lower per episode ($51,585 vs $56,865). Because these estimates are based on allowed amounts in Medicare claims, they reflect net spending across all services delivered during the episode, including outpatient wound care, hospitalizations, and complication-related care in 1:1 matched cohorts.
Scaled to the population level, these differences imply that for every 1,000 eligible Stage 3 PI episodes treated with CAMPs instead of SoC alone, total Medicare spending would be expected to decrease by approximately $3.8 million to $5.3 million, depending on episode duration. These reductions occurred alongside lower sepsis, fewer hospital-based encounters, and lower mortality, indicating that the economic impact is driven primarily by avoidance of high-cost downstream events rather than reductions in outpatient care intensity.
From a coverage-policy perspective, these findings are directly relevant to CMS decision-making in a high-risk population where access to advanced wound therapies varies across regions and care settings. In that context, the persistently low uptake of CAMPs across 2016–2024, only 10,864 of 110,381 eligible Stage 3 PI episodes (~9.8%), signals a potential access or implementation gap. A likely contributor is variation in coverage policies and documentation requirements across jurisdictions and wound etiologies. Future analyses should quantify PI-related CAMP utilization by MAC/jurisdiction and test whether policy differences are associated with utilization patterns and downstream outcomes.
Importantly, Stage 3 PIs carry substantial morbidity, frequent hospitalization, and excess mortality, making prevention of infection and inpatient escalation a central policy objective. Accordingly, evidence that CAMP-treated episodes are associated with fewer severe complications and lower overall spending supports consideration of clearer, nationally consistent coverage criteria for appropriately selected patients, paired with ongoing claims-based surveillance of sepsis, hospitalization, and mortality.
Contextualizing burden and trend relevance
These results are salient given evidence that PI burden in older adults has increased globally over time, and are especially relevant to Medicare populations, where frailty, immobility, and multimorbidity increase the likelihood that PI episodes will be complicated by infection and inpatient escalation. Because advanced-stage PIs are a major driver of national cost burden, strategies that reduce sepsis, hospitalization, and mortality could have outsized clinical and economic impact. Accordingly, sepsis, hospitalization, and mortality are appropriate patient-centered endpoints for evaluating the real-world value of advanced therapies in Stage 3 PI ulcers.
Limitations
Several limitations are inherent to administrative claims analyses: differences by site of service, limited non-facility sampling, inability to capture recurrence, heterogeneity in wound etiology heterogeneity, and lack of direct quality-of-life measurement. Consistency across the ≥90- and ≥120-day cohorts reduce the likelihood that results are driven by episode construction and supports generalizability to chronic Stage 3 PI treatment patterns. In addition, international consensus guidance notes ongoing variability in PI classification and documentation across care settings, which may contribute to diagnostic misclassification in administrative claims analyses.11 Separately, the “Regulatory and Reimbursement Reform for Cellular, Acellular, and Matrix-like Products (CAMPs)” consensus describes inconsistent audit and coverage interpretations that may influence real-world utilization and access patterns, which are not fully measurable in claims-based effectiveness analyses.12 Additional limitations include:
-
Residual confounding: even with matching, claims do not capture wound size, undermining extent, presence of nonviable tissue, offloading adherence, caregiver support, or staging validation and location at each encounter.
-
Outcome proxies: “healing” is not directly observable; utilization/cost signals may reflect both disease severity and care patterns.
-
Coding variability: diagnosis and procedure coding may vary by provider/system and over time.
-
Generalizability: this analysis focuses on a defined Stage 3 HOPD/inpatient subset of U.S. Medicare patients and may not be generalizable to a broader population.
-
Site-of-service sampling: This analysis used Medicare institutional claims files (Hospital Outpatient Department and Inpatient) to construct treatment episodes and evaluate outcomes. The Medicare Carrier file was not included because only a 5% sample was available in the dataset; inclusion would have substantially reduced the matched cohort size (from approximately 7000 episodes per group to roughly 350 per group), thereby limiting statistical power. Accordingly, the observed CAMP utilization reflects institutional-site claims and may not fully capture product-selection patterns across all sites of care.
Implications
In a high-risk Medicare Stage 3 PI population, CAMP use in real-world practice was associated with lower mortality and fewer severe complications alongside lower total spending, suggesting potential value in avoided hospital resource utilization. These findings support further work to incorporate greater clinical granularity (e.g., wound dimensions, infection confirmation), evaluate recurrence and durable closure, and assess heterogeneity across anatomic site and comorbidity strata.
Conclusion
In matched Medicare Stage 3 PI episodes (2016–2024), treatment with CAMPs plus SOC was associated with lower sepsis, lower acute-care utilization, lower major amputation, and lower mortality, with lower total and PI ulcer coded spending, despite longer episode duration and greater outpatient visit intensity. These results suggest that, in real-world practice, CAMPs may help reduce severe complications and costly inpatient escalation in Stage 3 PIs, warranting confirmatory studies with richer clinical detail and direct healing endpoints.